Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 31.07.2025
Medela Thopaz+ Product Documentation Analysis Report
Overview and Product Description
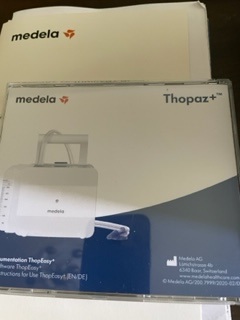
The image shows the documentation and software package for the Medela brand’s Thopaz+ digital drainage system. The product consists of a CD/DVD case and an additional printed document. According to the image on the packaging, the Thopaz+ device to which this documentation belongs is a medical aspiration and drainage unit. The examined items include the user manual and related software for this device.
- Product Type: Medical Device Documentation and Software
- Area of Use: Contains the necessary information and software for the use, installation, and management of the Medela Thopaz+ digital drainage system.
- Quantity Information: A total of 2 product components are visible in the image. One is a CD/DVD case in its transparent cellophane, and the other is a printed document seen in the background.
Brand and Model Information
The brand and associated model information of the products in the image are specified below.
- Brand: Medela
- Associated Device Model: Thopaz+
- Documentation/Software Name: ThopEasy+
Physical Condition and Content
The products appear to be in new or like-new condition. The CD/DVD case is clean and undamaged. No scratches, cracks, color fading, or dirt have been detected. The printed document in the background is also in a clean and proper condition. It is assessed that the products are generally unused.
Label and Code Information
The label information on the CD/DVD case is very clear. This information provides detailed information about the product’s identity and content. The information on the label is as follows:
- Content Title: Documentation ThopEasy+ / Software ThopEasy+
- Language of Use: There is an indication that the instructions are in English [EN] and German [DE].
- Manufacturer Information: Medela AG, Lättichstrasse 4b, 6340 Baar, Switzerland
- Web Address: www.medelahealthcare.com
- Reference Code: At the bottom of the label, there is a code in the format © Medela AG/200.7999/2020-02/B. This code indicates the document’s version and publication date.
- Production/Publication Date: The “2020-02” notation within the code suggests that this documentation was prepared or printed in February 2020.
Potential Risk Assessment
No physical deformation, wear, or damage has been observed in the documentation package examined in the images. The condition of the products is very good, and they do not pose any potential risk of malfunction related to their condition.