Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 28.01.2026
RESMED ResControl II Device and Accessories Analysis Report
Device Identification
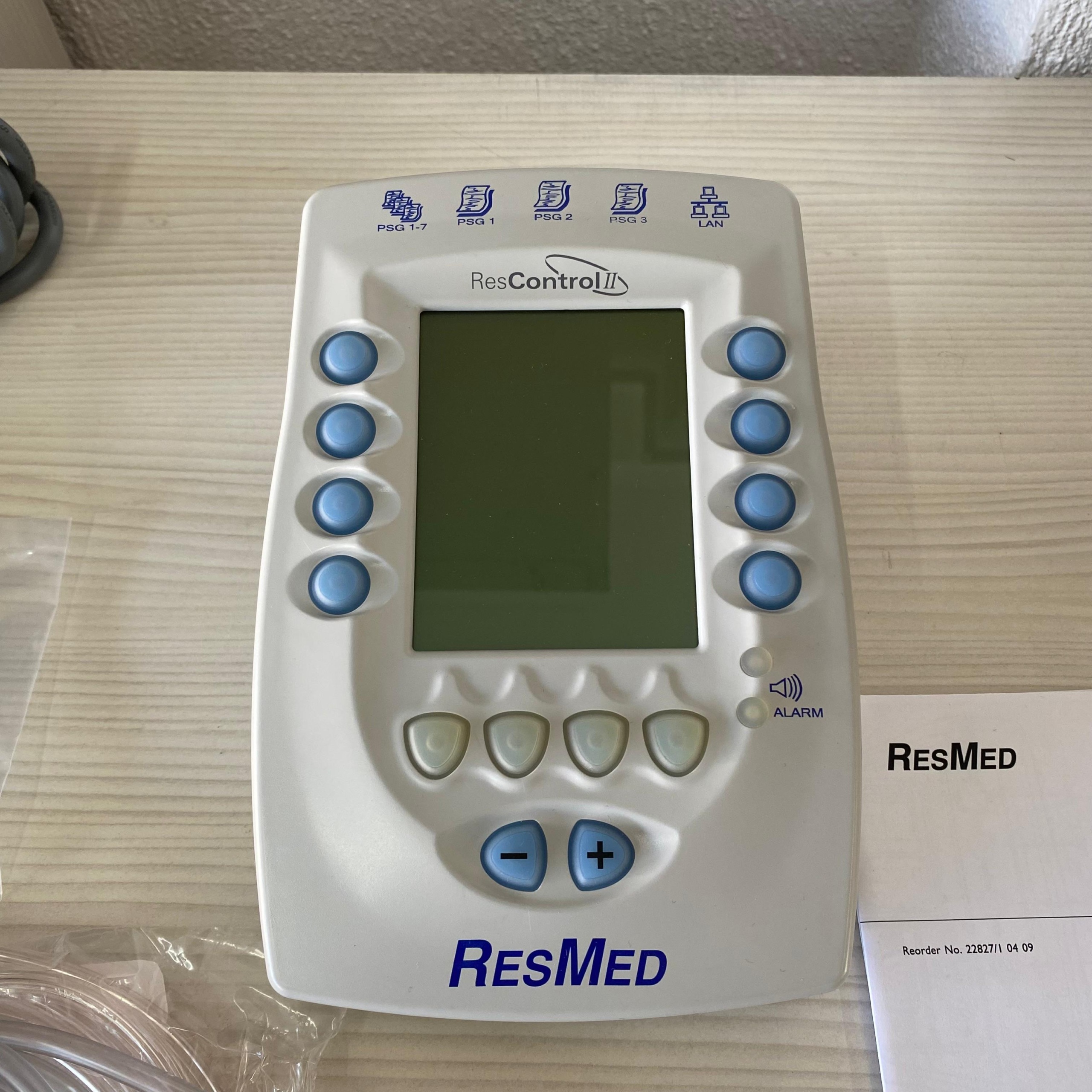
The examined images reveal a data communication and monitoring module identified for medical use. The device’s front interface, screen structure, and rear connection ports indicate that this product is a professional control unit designed to work integrated with respiratory devices or sleep laboratory equipment (Polysomnography – PSG).
Brand and Model
A detailed examination of the writings and logos on the product clearly identifies the device’s brand as the world-renowned medical device manufacturer RESMED, and its model as ResControl II. This information is clearly legible on the device’s front panel and the user manual cover.
Originality
The product’s plastic injection quality, corporate logo prints, orderly placement of ports, and the accompanying original user manual (Quick Reference Guide) prove that the device is an original RESMED production. No indications of aftermarket or replica parts were found.
Areas of Use
Considering the inputs located on the rear ports of the device in the images and the “PSG” notation on the front panel, it is understood that this device is used to convert data (pressure, flow, etc.) from RESMED brand respiratory therapy devices into analog signals for transmission to Polysomnography (PSG) systems or to transmit data over a local area network (LAN). It acts as a bridge between the therapy device and the recording system in sleep laboratories or advanced patient monitoring processes.
Quantity Information
The set shown in the images consists of the following parts:
- 1 RESMED ResControl II main unit
- 1 Gray power cable
- 1 Gray data connection cable (one end with a device-specific connector)
- 1 Air/pressure tubing (Nasal cannula or pressure line) in its original transparent packaging
- 1 Metal connection adapter/connector in a small pouch
- 1 User manual titled “ResControl II – Quick Reference Guide”
General Condition and Cosmetic State
The device’s cosmetic condition is at an excellent level. No yellowing, stains, or significant dirt are observed on the white/gray outer casing. The absence of color changes that might occur over years suggests that the device has either been used very little or has been very well preserved (e.g., stored in its box). There are no dents, cracks, or deep scratches.
Physical Deformation and Screen
The device’s casing is intact; there are no openings at the joints. The screen surface is clean, and no visible scratches or dead pixels were detected on the screen (physical surface analysis was performed as the device was off). The keypad immediately below the screen and the blue control knobs on the right/left sides appear sturdy and clean. There is no fading of text and icon prints.
Mechanical and Electronic Components
Upon examining the rear panel, which is seen in detail in Image 3:
- Ports: All socket inputs (LAN, Serial Ports, Pneumatic inputs) are free from rust, and their pins are straight and clean.
- Air Inlets: The 3 metal air/pressure nozzles (likely labeled PSG 1, PSG 2, PSG 3) retain their shine; there is no oxidation.
- Ventilation: No dust accumulation is observed on the side ventilation grilles.
- Switch: The On/Off (I/O) switch is physically intact.
Accessories
The accessories included with the set are:
- Manual: The “Quick Reference Guide” is very clean, neither worn nor torn. The cover reads “Reorder No: 22827/1 04 09”.
- Tubing: The transparent tubing set is in its original sealed/locked pouch and appears unopened.
- Cables: There is no peeling, breakage, or crushing of the insulation on the power and data cables. They are neatly stored with cable ties.
Label and Date Information
The main label containing the device’s serial number (usually found on the underside of the device) is not visible in the images. However, the following codes are available:
- Booklet Code: 22827/1 04 09 (This code indicates that the booklet was printed in April 2009 or its revision date. It can be inferred that the device was manufactured after this date.)
Potential Failure Risk
As a result of the visual analysis, no risk factors such as burn marks, liquid contact, severe impact, or oxidation were observed on the device. The cleanliness of the ports and the “like new” overall condition suggest a low risk of potential hardware failure. Although the product’s functionality can only be tested when powered on, its physical condition indicates that the device has been stored in a healthy environment.