Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 16.10.2025
Cynosure Icon Aesthetic Laser Device Analysis Report
Overview and Device Description
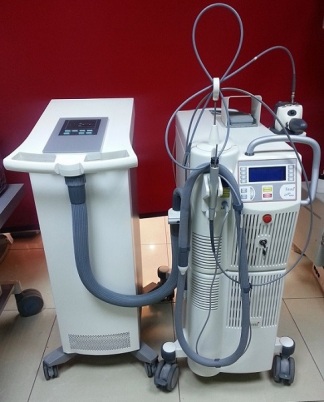
The device shown in the visuals is a laser system used in professional aesthetic and cosmetic applications. The general design of the device consists of a wheeled main unit and application handpieces connected to this unit. It is understood that the product is used in various dermatological procedures such as skin rejuvenation, spot treatment, and hair removal. The analysis was conducted based on one main unit and at least one application handpiece.
Brand and Model
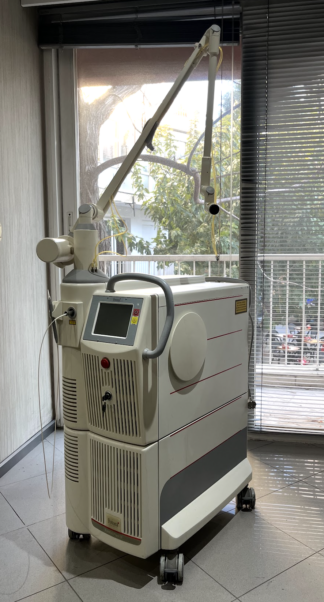
A detailed examination of the visuals clearly shows the device bears the Cynosure brand and Icon model. This information is present on both the device’s casing and the touchscreen interface.
Condition and Status Assessment
The general condition of the device is assessed as used. Its casing is generally clean and has a well-maintained appearance. The craftsmanship, logo placement, and quality of the screen interface indicate that the device is original. No serious physical deformation has been observed.
- Physical Deformation: The device’s white plastic casing shows no prominent large scratches, cracks, dents, or discoloration. Only minimal wear due to use might be present.
- Cleanliness: The general surfaces of the device and the ventilation grille appear clean, with no heavy dust or dirt accumulation.
Mechanical and Electronic Components
The mechanical and electronic components of the device appear to be in good condition, as understood from the visuals.
- Mechanical Parts: There is no visible damage, breakage, or disconnection in the cradle where the application handpiece rests, the handpiece itself, or the cable/hose system connected to the device. The ventilation grille located at the bottom is intact.
- Electronic Parts: The device’s touchscreen is operational and displays an image. No cracks, fractures, or abnormal color distribution have been observed on the screen.
Screen Analysis
The device’s touchscreen is active and displays a system interface. The screen shows “Icon Aesthetic System” and a list indicating the system’s treatment records or patient data. This list includes dates and various numerical data. The clarity and readability of the interface confirm that the screen is functional.
Accessories
In the visual, there is one application handpiece connected to the device’s main unit. The handpiece is attached to the device with a flexible cable and rests in its cradle. The power cable, necessary for the device’s operation, is also visible in the background of the visual. Other accessories such as a box, additional handpieces, or a user manual are not present in the visual.
Technical Information and Labels
No label or plate containing specific technical information such as serial number, REF code, lot number, or year of manufacture is legibly present on the device in the available visuals.
Malfunction Status and Potential Risks
Based on visual inspection, there is no evidence of an existing malfunction in the device. There is no error message on the screen, and no damage is visible on the device’s casing. No significant defects that could pose a potential risk of malfunction, such as crushed cables, rust on the casing, or severe wear, have been observed. The current condition of the device suggests it is suitable for regular use.