Ready For Sale
Secondhand Dana Magnum Q Switch Nd YAG Aesthetic Laser Device
Price: USD$ 6.000,00 Approx: 270.000,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 08.04.2026
Report code: 1775655162
Dana Magnum Nd:YAG Laser Device Analysis Report
Device Identification and Basic Information
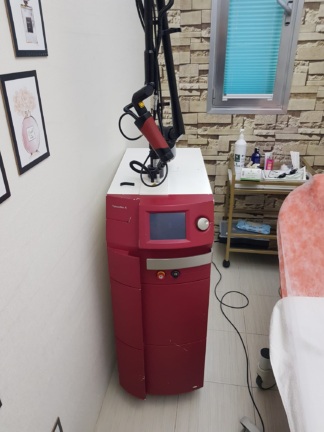
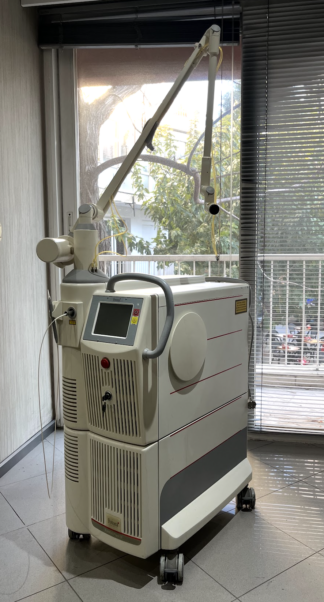
The product in the images is a wheeled and stand-mounted medical/aesthetic laser device. On the front body of the device, the text Dana Magnum is clearly visible, and on the side body, the phrase ND:YAG LASER SYSTEM is clearly legible. Accordingly, the brand of the device has been identified as Dana, and the model as Magnum.
Areas of Use
Nd:YAG laser systems are used in dermatology and medical aesthetic clinics. These devices are generally clinical-type equipment suitable for procedures such as tattoo removal, skin rejuvenation, and pigmentation (spot) treatment.
Quantity Information and Originality Assessment
- Quantity: The images show 1 main device and 1 articulated (multi-jointed) treatment arm integrated into the device.
- Originality: Upon examining the device’s casing structure, the mechanical design of the articulated arm, the logo prints on the body, and the industrial connection points on the rear panel, the product is assessed to be an original device.
General Condition and Physical Deformation
The device is in used condition. Various areas of the white outer casing, especially on the side panels and the lower parts of the handle, show superficial dirt, dust, and minor scuff marks. No cracks, fractures, or deep dents have been observed on the casing. The grey panel at the rear appears clean and physically sound.
Mechanical and Electronic Component Analysis
- Mechanical Components: The 4 wheels that enable the device to move on the floor appear sound. The push and pull handle at the front is intact. The joints and physical integrity of the metallic articulated arm that directs the laser beam are preserved.
- Electronic Components and Connections: On the front panel, there is a key-operated switch used to start the device, with the key inserted. On the rear panel, there is a power cable input, a circular connection socket, and an additional socket input at the bottom. The black ventilation grilles on the rear panel are open and show no physical damage.
Screen Condition
The main control screen is located on the front upper part of the device. The screen is in the off position, and no cracks or deep scratches are visible on its surface. On the rear panel of the device, there is a small LED indicator panel displaying red digital numbers.
Accessories
On the top part of the device, there is a black cable or pedal accessory wrapped in transparent nylon. A thin cable line leading to the firing mechanism is present on the articulated arm. External accessories such as a power cable or protective goggles belonging to the device are not clearly visible in the images.
Label and Document Information
No information label, invoice, or warranty document indicating the device’s year of manufacture, serial number, lot number, or REF code could be identified in the images.
Malfunction and Risk Assessment
No clear break, severed cable, or error message that would prevent the device from operating has been identified in the images. The general condition of the device is good. The observed superficial dirt and scratches are cosmetic and do not pose a potential risk of malfunction.