Ready For Sale
Secondhand HHP Andumedic 3 Pro Andullation Therapy Device
Price: USD$ 2.200,00 Approx: 99.000,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 10.04.2026
Report code: 1775829897
HHP Andumedic 3 Professional Therapy Device Analysis Report
Device Identification and Basic Information
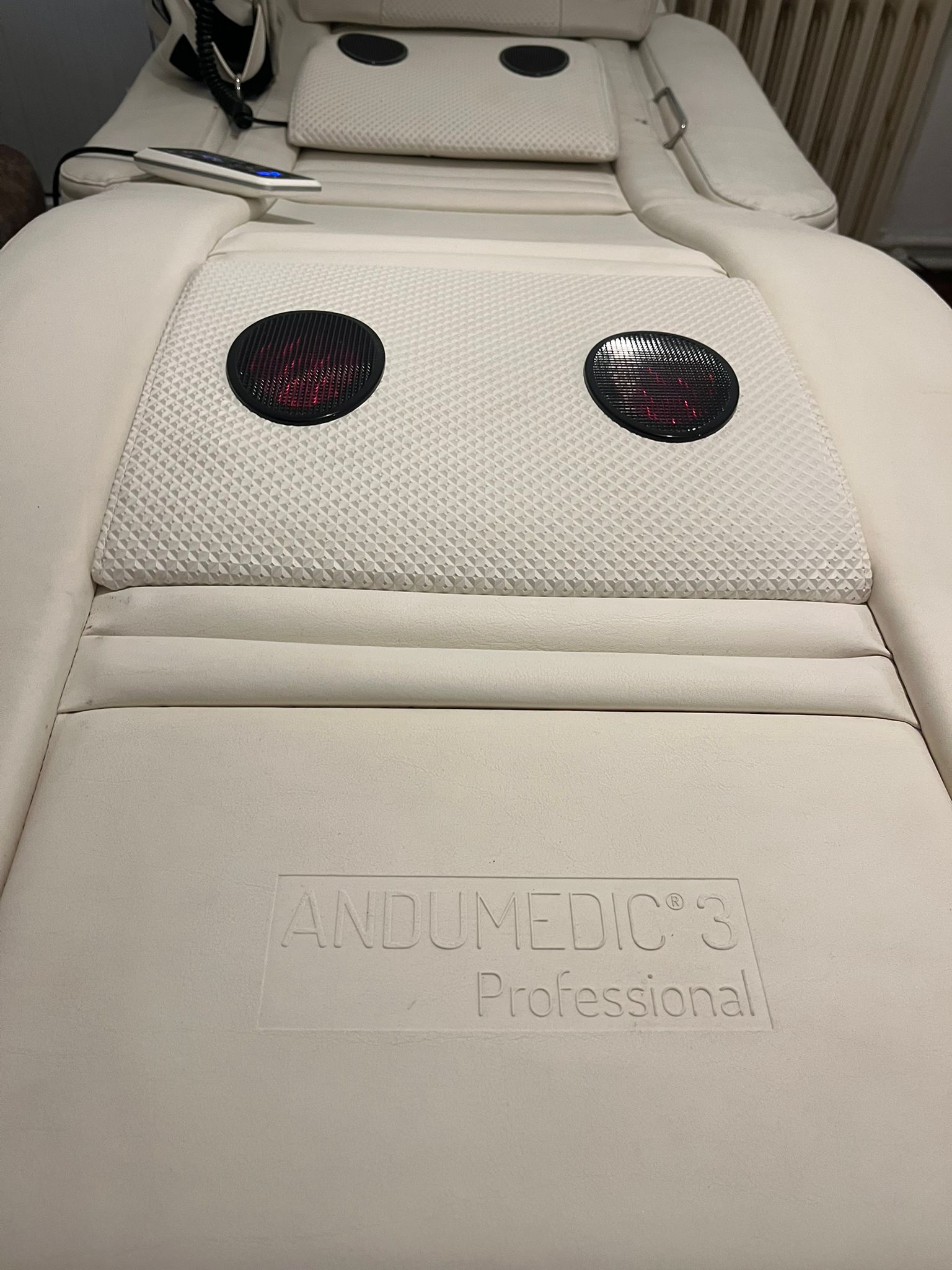
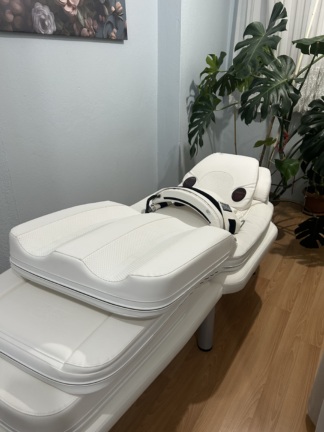
The product in the images is a therapy bed system designed for holistic body therapy, massage, and andullation applications. The hhp logo is embossed on the head section of the device. In the lower section towards the foot, the text ANDUMEDIC 3 Professional is clearly legible. Accordingly, the brand of the device has been identified as HHP and the model as Andumedic 3 Professional.
Authenticity and General Condition
Considering the craftsmanship of the embossed logos, the quality of the stitching, the texture of the synthetic leather material used, and the integration of the infrared light panels, the product is assessed to be authentic. In general terms, the device appears to have been used but is very well-maintained, clean, and in high condition.
Areas of Use
This device is used for physical therapy, rehabilitation, pain management, and general relaxation. It operates on a therapy principle that offers a combination of vibration and infrared heat. It is suitable for physical therapy centers, sports clubs, beauty and health centers, and individual home use. It is sized appropriately for adult human anatomy.
Physical and Mechanical Condition
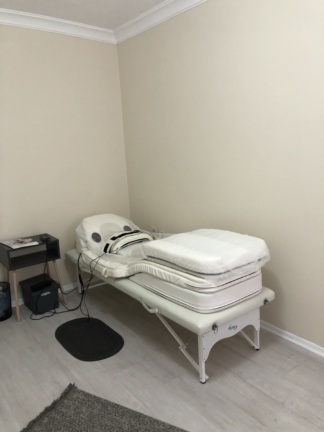
No tears, holes, abrasions, color fading, or stains have been observed on the white synthetic leather material covering the outer surface of the device. The seams are intact. The metal support frame on which the device is placed appears stable. There are no visible mechanical issues or deformations at the folding points and joints.
Electronic Components and Accessories
The device features red infrared light panels integrated into circular, honeycomb-textured surfaces that provide heat during therapy. It is observed in the images that these panels are actively lit and functioning. The hardware and accessories identified for the device are as follows:
- One main therapy bed unit
- One wired hand controller
- One velcro fastening and therapy belt
- One head and neck support cushion
- One metal support stand
The wired hand controller has a digital screen and control buttons. However, due to the angle and resolution of the images, the text or menu contents on the screen cannot be read. There are no signs of crushing or breakage on the controller cable or the connection points entering the device.
Label Information and Fault Status
There are no technical labels, barcodes, or information plates visible in the images showing the device’s serial number, production year, lot number, or REF code. Likewise, documents such as invoices, warranty certificates, or user manuals are not present in the images.
No visible existing faults, broken parts, or missing equipment have been detected on the device. The device is in very good condition regarding potential fault risks. Only normal wear and tear can be expected in the bending points of the hand controller cable and areas of the synthetic leather surface subject to intense friction during long-term use, but no immediate risk in this regard has been observed.