Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 16.02.2026
Asclepion MeDioStar Laser Device Analysis Report
Device Identification
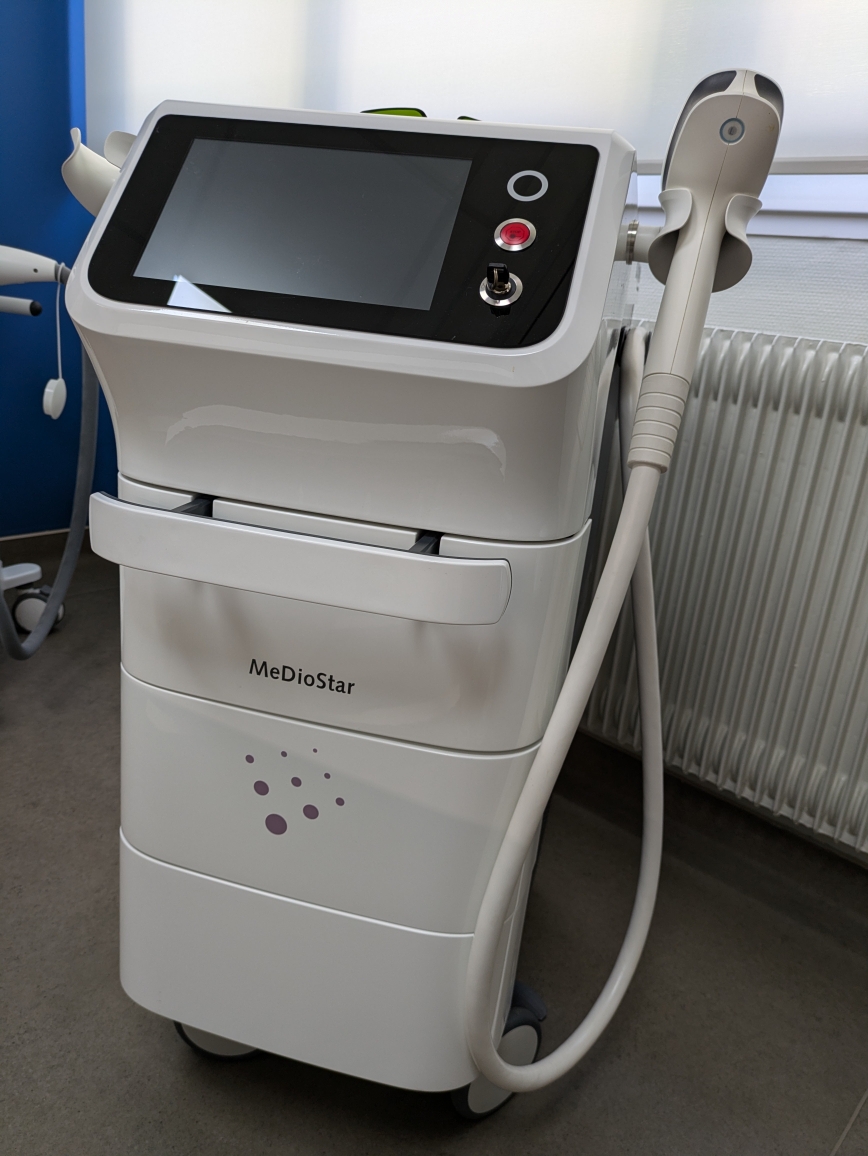
The product analyzed in the visuals is a medical aesthetic laser device intended for professional use, featuring a wheeled stand structure. The device consists of a main body, a large touchscreen integrated into this body, a movable wheeled system, and a wired handpiece used for treatment applications. Labels and connection points on the device’s rear panel confirm that it is a medical-grade electronic device.
Brand and Model
The inscriptions and labels on the product have been examined in detail:
- Device Brand and Model: The inscription “MeDioStar” is clearly visible on the front of the device, and “Asclepion Laser Technologies GmbH” is stated as the manufacturer on the rear information label, with “MeDioStar” again as the model name. The brand logo, a purple dotted pattern, is present on the front panel.
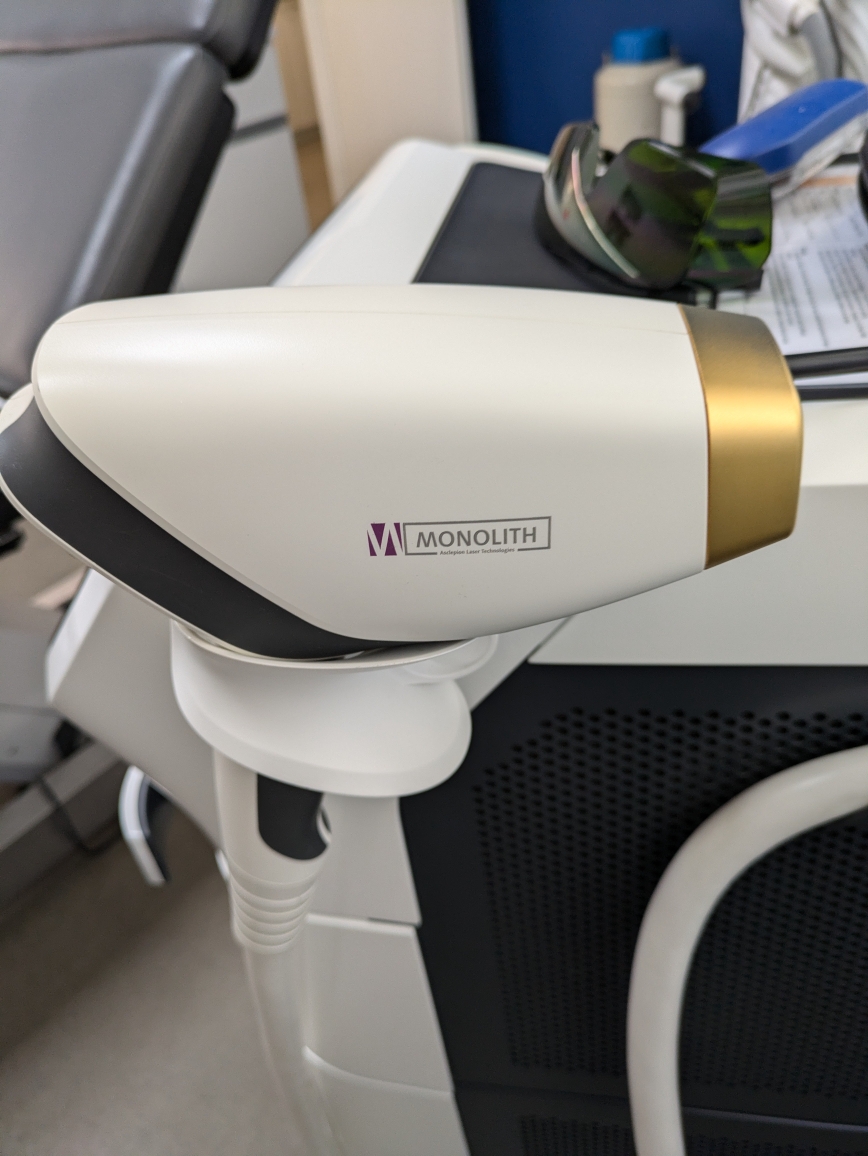
- Handpiece Model: The inscription “MONOLITH” and “Asclepion Laser Technologies” below it are clearly visible on the handpiece section of the device. The gold-colored tip and ergonomic design of the handpiece reflect the characteristic features of this model.
Authenticity
Considering the print quality of the labels on the device, logo placements, the official type plate (silver-colored plate) on the rear panel, the “CE 0123” certification mark, and the overall manufacturing quality, the product is assessed to be an original production of the Asclepion brand.
Areas of Use
Asclepion MeDioStar is a high-tech diode laser device used in dermatology clinics and aesthetic centers. Its primary areas of use include:
- Laser hair removal (permanent reduction of unwanted hair).
- Vascular treatments (capillary vessel treatments).
- Skin rejuvenation and acne treatment applications.
- The Monolith handpiece is an advanced accessory offering faster and more comfortable application.
Quantity Information
The following parts were identified in the visuals:
- 1 Asclepion MeDioStar main device.
- 1 Monolith laser application handpiece (with integrated cable to the device).
- 1 Laser protective eyewear (partially visible on the tray behind the device in Visual 2).
General Condition
The cosmetic condition of the device is visually excellent. There is no yellowing, heavy dirt, or signs of use on its outer casing. It gives the impression of being “used but very clean and well-maintained” or “refurbished.” The device’s white color retains its brightness.
Physical Deformation
No dents, deep scratches, cracks, or dings were detected on the visible surfaces of the device (front panel, side covers, rear metal panel). Plastic components are intact and in place. There are no signs of wear on the screen bezel or handles.
Mechanical Components
The wheels, handpiece holder, handles, and key-operated lock mechanism appear intact. No breakage, crushing, or peeling is observed on the thick white hose (fiber optic/power cable housing) connecting the handpiece to the device; the hose maintains its proper form.
Electronic Components
As the device is in the off position, electronic functions could not be tested. However:
- The “Emergency Stop” button on the front panel is intact and in place.
- The key is inserted into the key switch, and the lock mechanism is complete.
- No physical cracks or breaks are visible on the touchscreen.
Label Information
The technical data read from the silver label on the rear of the device is as follows:
- Manufacturer: Asclepion Laser Technologies GmbH (Jena, Germany)
- REF (Reference Code): M10-0100-000
- SN (Serial Number): M100100226
- Manufacturing Date: 2019-11 (November 2019)
- Power Input: 100-120 V ~ / 220-240 V ~, 50/60Hz, 1500VA
- Weight Information: 45 (65) kg (Likely empty and fluid-filled weights)
- Temperature Range: 15°C – 30°C
Maintenance and Inspection Information
A circular orange technical service/calibration label is located on the rear bottom part of the device. The label features the “TechniLase” logo and highlights the year “2025.” This indicates that the device’s periodic maintenance or electrical safety tests are scheduled for 2025 or are valid until that year. It is an important sign that the device is professionally tracked.
Accessories
In addition to the main device, the “Monolith” brand application handpiece is shown in detail in the visual. The contact tip of the handpiece appears clean, and there is no peeling on its gold-colored coating. Furthermore, one protective laser goggle (with dark green lenses), likely for the operator or patient, is present on the device’s rear tray.
Battery Status
As this device is a high-power laser system, it operates not with an internal battery, but with mains electricity (220-240V). The label specifies a power consumption of 1500VA. A portable battery system is not visible in the visual.
Screen Analysis
At the top of the device, there is a considerably large touchscreen LCD panel, likely 10 inches or more in size. As the screen is currently off (black), the software interface and operating hour information cannot be read. However, the screen surface is clean and smooth.
Year of Manufacture
As a result of the label analysis, it has been definitively determined that the device was manufactured in the 11th month of 2019.
Potential Malfunction Risk
Based on the physical examination in the visuals:
- The device’s condition is very good and does not pose any significant risk factors.
- As cable and hose connections appear healthy, the risk of fiber optic transmission breakage seems low.
- Since the device likely operates with a water cooling system (the 45/65kg weight difference on the label indicates a water reservoir), draining the water during transport is critically important.
Conclusion Comment
The examined Asclepion MeDioStar, despite being manufactured in late 2019, is a device that has been used extremely meticulously and well-preserved. Its possession of Monolith handpiece technology indicates a current configuration. The service label bearing “2025” gives the impression that the device’s maintenance history is recorded and regular. Cosmetically and structurally, it appears complete, clean, and ready for professional use.