Ready For Sale
Secondhand GL090 Diode Laser Epilation Device
Price: USD$ 4.200,00 Approx: 189.000,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 21.02.2026
Diode Laser Hair Removal Machine Analysis Report
Device Detection and Identification
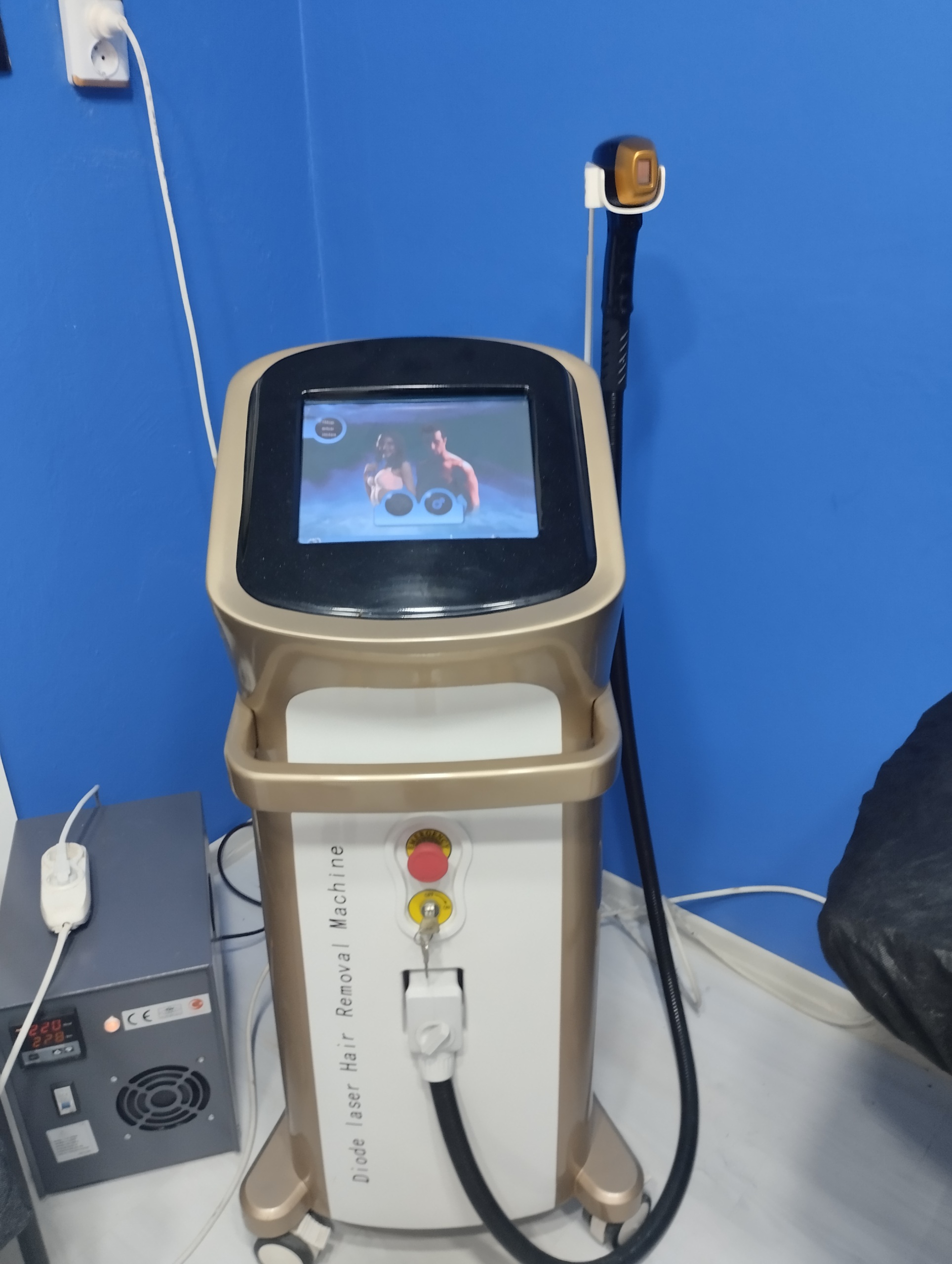
The examined visuals belong to a tower-type (vertical) laser hair removal device, designed for use in professional aesthetic and beauty centers. The general structure of the device consists of a touchscreen main control unit, a handpiece, and a wheeled body. Additionally, on the left side of the device, there is a gray box with cooling grilles, presumed to be an external power supply or voltage regulator. The main function of the device is laser hair removal, applicable to different skin types.
Brand and Model Information
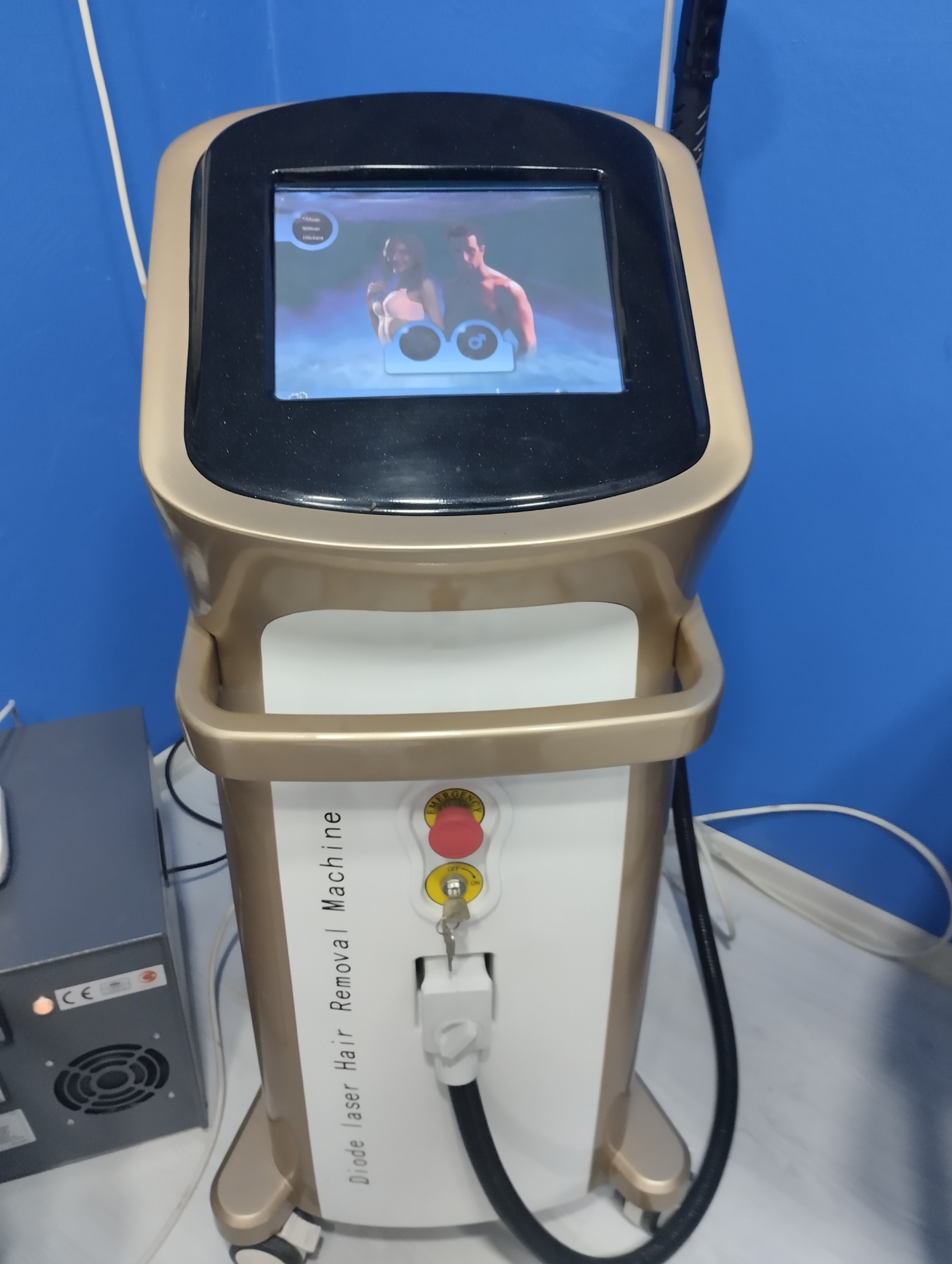
The phrase “Diode laser Hair Removal Machine”, written vertically on the front body of the device, is clearly visible. A detailed examination of the visuals did not reveal a specific brand logo (such as Alma, Candela, Lutronic) or a distinct model number belonging to the manufacturer. This phrase is typically seen on devices that are generically produced or marketed as OEM (Original Equipment Manufacturer) and can be named by importer companies. Since no other data confirming the brand 100% was found on the device’s interface or casing, the device should be considered an unnamed model with “Diode Laser” technology.
Screen Analysis and Technical Data
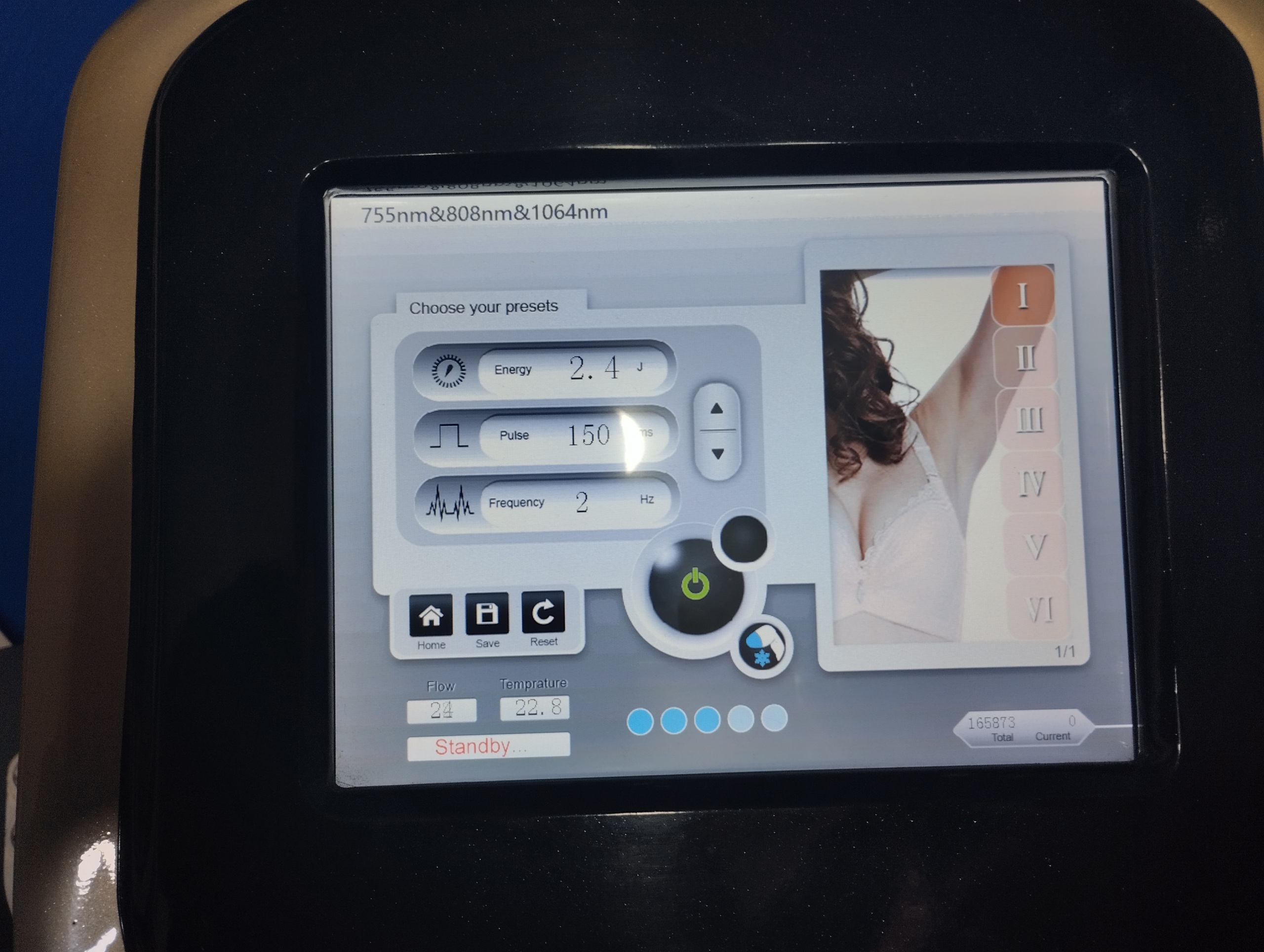
A detailed analysis of the device’s touchscreen revealed critical technical data about its operational capabilities:
- Wavelength Technology: The phrase “755nm&808nm&1064nm” at the top of the screen indicates that the device uses hybrid or triple wavelength technology (simulating Alexandrite, Diode, and Nd:YAG wavelengths). This feature indicates that the device caters to different hair and skin types.
- Skin Type Scale: The Fitzpatrick skin type classification, ranging from I to VI, is present on the right side of the screen. This indicates that the device can perform procedures on a wide range of skin tones, from fair-skinned (Type I) to dark-skinned (Type VI).
- Parameter Settings: The analyzed screen image shows that the energy level is set to 2.4 J (Joule), the pulse duration to 150 ms, and the frequency to 2 Hz.
- System Status: The device is in “Standby…” mode. The Flow value is read as 24, and the Temperature value as 22.8 degrees, indicating that the cooling system is active and sensors are reading data.
Usage Duration and Shot Count
The most critical section of this report for the recipient is the counter information indicating the device’s operational life. A clear number is displayed under the “Total” heading in the lower right corner of the device’s screen:
- Total Shot Count (Total Counter): 165873
This figure documents that the device’s laser handpiece has fired a total of 165,873 shots. This information is vital for assessing the handpiece’s lifespan and remaining usage capacity. Furthermore, the number “165873” seen on the main screen also confirms this counter.
Areas of Use
The device is used in aesthetic clinics, beauty centers, and dermatology polyclinics for the permanent reduction of unwanted hair (laser hair removal). Thanks to its multiple wavelength technology, it is suitable for application on fine/thick hair structures and various skin tones, including tanned skin.
Accessories and Components
The components identified with the device in the visuals are as follows:
- 1 Laser Hair Removal Handpiece and its hose.
- 1 Device Key (inserted into the ignition).
- 1 External Power Unit/Regulator (bottom left, gray metal box, with digital display and CE mark).
- Connection cables located at the back of the device.
General Condition and Physical Condition
The device’s outer casing consists of a combination of “Gold” (golden yellow) and white colors. No visible cracks, deep scratches, or color fading were detected on the plastic parts. The device’s paint appears vibrant and clean. The handpiece hose is not bent and appears sturdy. The “Emergency” button on the device is in place and intact.
Mechanical and Electronic Component Assessment
Electronic Status: The device’s LCD screen is working flawlessly; no dead pixels, lines, or darkening are observed. It is understood from the screen images that the software is loaded, menus can be navigated, and sensors (temperature and water flow sensors) are reading values.
Mechanical Status: Although the condition of the wheels cannot be fully discerned from the visuals, the tower structure stands balanced. The ignition key is in its slot. The handpiece holder is sturdy. The voltage indicators on the external power unit on the side are active (the red LED indicator shows “228” or a similar voltage value), which proves that electricity is flowing.
Authenticity Assessment
The product appears to be a generic device of Chinese origin. Since it does not have the logo of a major internationally recognized medical company, it should be categorized as “unbranded/imported.” Although a “CE” mark is visible on the adjacent power regulator, this mark alone does not guarantee full compliance of the entire device with European standards; the craftsmanship is of standard quality.
Potential Risks and Considerations
The device appears to be in working condition and clean. However, technical service and spare part procurement for “no-name” (unbranded) devices may differ compared to branded devices. The lifespan of the laser handpiece (diode bar) is a consumable material; having made 165,873 shots indicates that the handpiece has been used, but a definitive remaining lifespan cannot be assigned without manufacturer data, as the total lifespan of the handpiece (e.g., 5 million, 10 million, 20 million shots) will vary depending on its quality. Currently, there are no malfunction warnings or signs of physical damage.