Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 23.02.2026
Diode Laser Hair Removal Machine Analysis Report
Device Identification
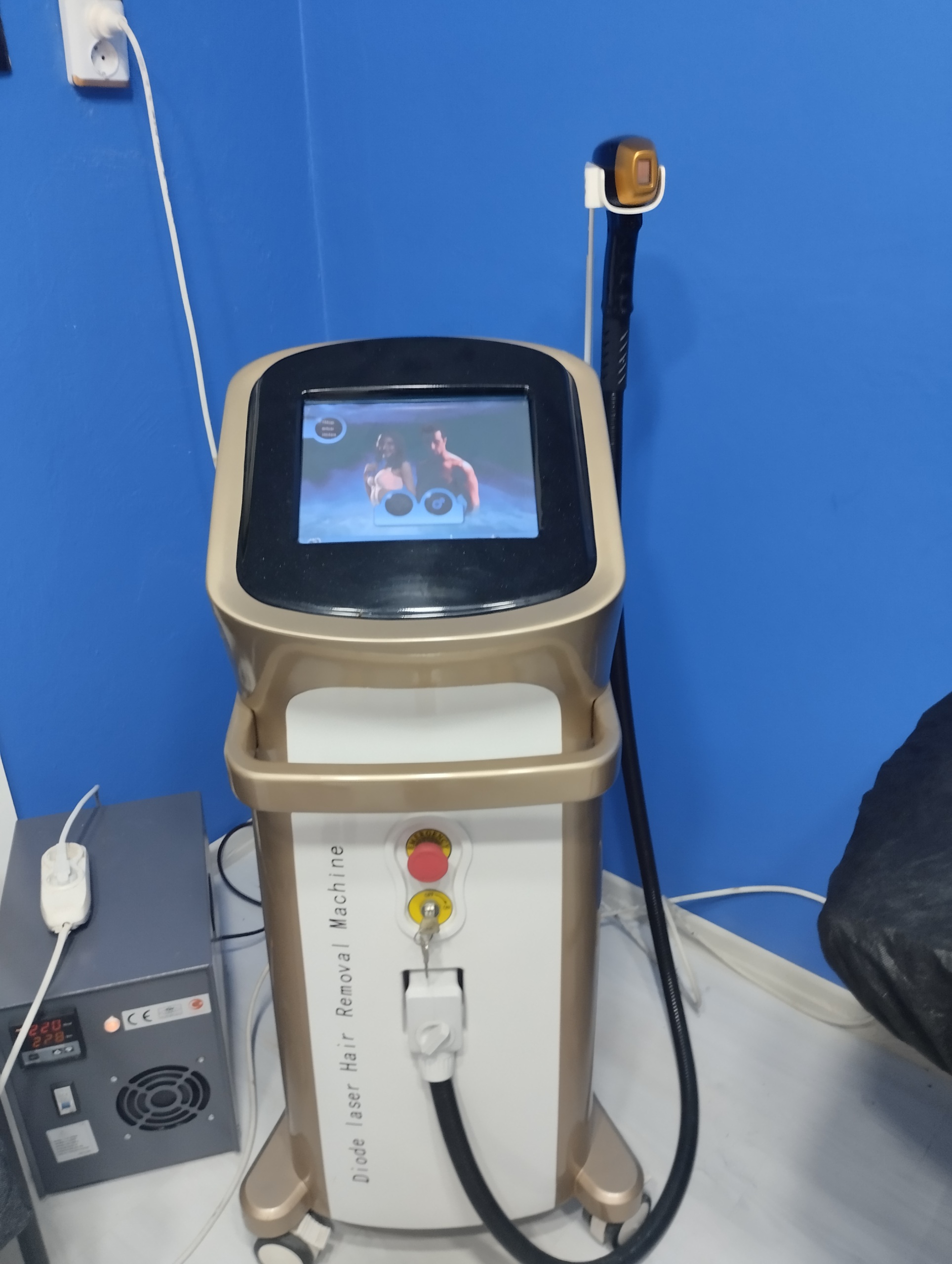
The examined device is a tower-type (vertically rising from the floor) professional laser hair removal machine used in dermatology and medical aesthetics for the permanent removal of unwanted hair. The system consists of a main body unit, a control screen, and an application handpiece.
Brand and Model Information
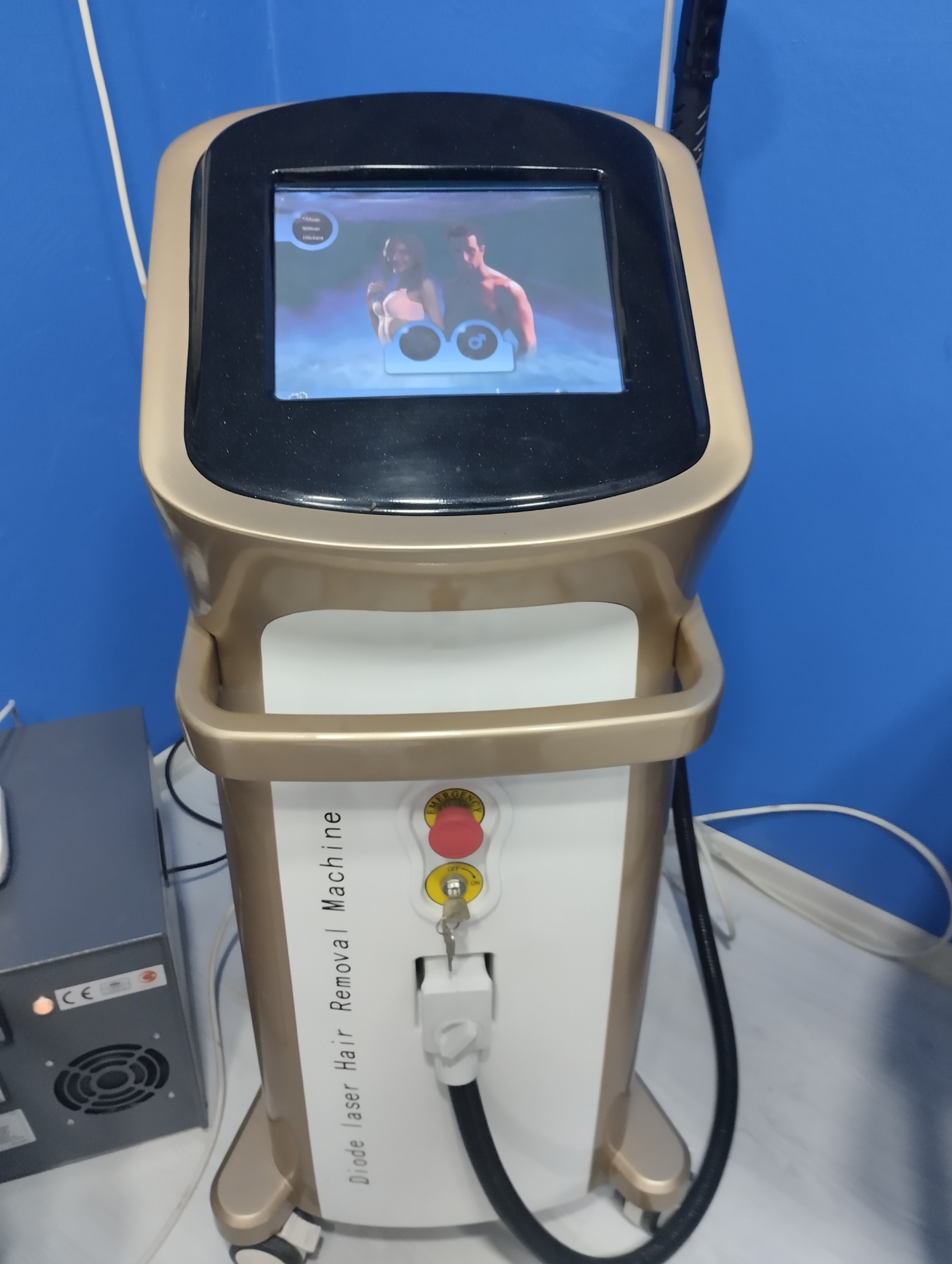
During a detailed examination of the device’s body, screen, and software, no specific brand logo, model name, or type number belonging to the manufacturer (e.g., Alma, Candela, Cynosure, etc.) could be visually identified. On the front of the device, on the left side, the phrase “Diode laser Hair Removal Machine” is vertically displayed. The product exhibits generic (OEM) manufacturing characteristics.
Originality and Origin
No distinctive serial number label, embossed logo, or registered trademark of a renowned brand is visible on the product. The interface software and casing design exhibit a common design language found in Far Eastern (imported) generic diode laser devices.
Technical Data and Operating Principle
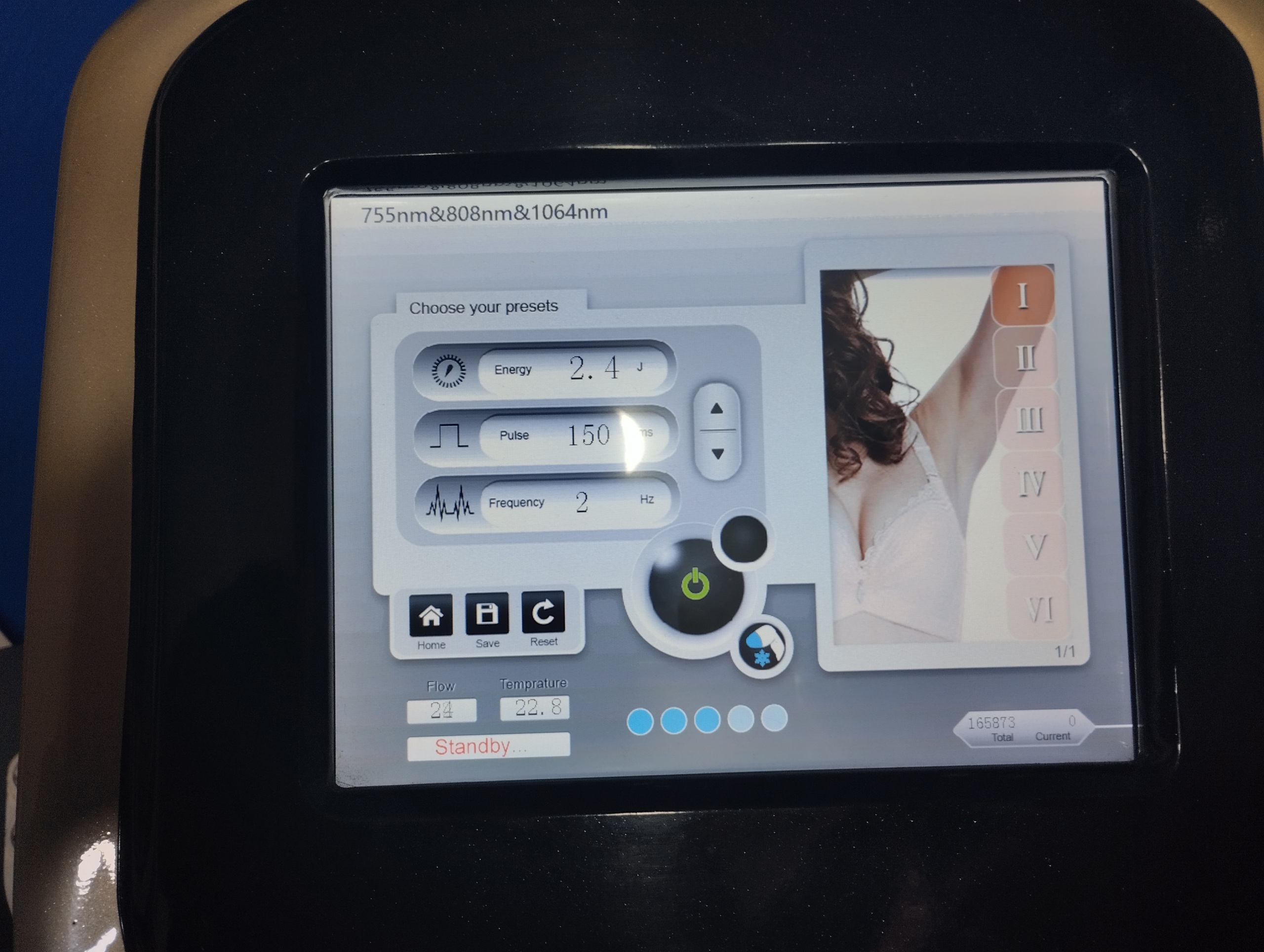
Based on the data obtained from the device’s screen interface, the system appears to have hybrid wavelength technology. The upper panel displays the phrase “755nm&808nm&1064nm”. This indicates that the device operates by combining or supports three different wavelengths (Alexandrite – 755nm, Diode – 808nm, Nd:YAG – 1064nm).
Screen Analysis and Interface Parameters
The device has a large touch-sensitive LCD panel, and at the time of the image, the device is operational (active). The instant parameters and information read on the screen are as follows:
- Operation Mode: “Standby…”
- Energy Setting: 2.4 J (Joule).
- Pulse (Pulse Width): 150 ms.
- Frequency (Speed): 2 Hz.
- Skin Type Scale: Fitzpatrick skin type selection buttons from I to VI are available.
- Gender Selection: Selection can be made using Male and Female figures.
- System Information: A software/ID code “7da018401847da” is located in the upper left corner.
- Language Option: Language selection is available with English and Russian flags.
- Cooling and Flow Sensors: “Flow”: 24 units, “Temperature”: 22.8 °C are reported.
Service Life and Counter Information
The counter information showing the device’s total shot count is clearly readable on the operation screen. This data is critical for estimating the remaining life of the diode bars.
- Total Shots (Total): 165873
- Current Session (Current): 0
On the login screen, the numbers “1234” are visible, likely a password entry or another counter value. However, the main usage counter is 165,873.
General Condition and Physical State
The device’s outer casing consists of white and gold-colored plastic/composite panels. The general condition analysis based on visual inspection is as follows:
- Casing Integrity: No cracks, fractures, or deep dents are visible on the front panel, side covers, or handle bars. Casing joints appear regular.
- Paint/Surface Condition: There are no signs of significant discoloration, peeling, or yellowing. The cosmetic condition is good.
- Screen: The touch screen’s brightness is adequate, with no pixel loss or impact marks. Text is clearly readable.
Mechanical and Safety Components
- Emergency Stop Button: A red “Emergency” button is present in the center of the front panel and appears physically sound.
- Key Switch: The power on/off switch is in place, and its key is inserted. The labels around the switch (“OFF”, “ON”) are readable.
- Handpiece Connection: The white connection socket (with locking mechanism) at the front bottom is securely seated.
Accessories and Additional Equipment
- Laser Handpiece: One laser application handpiece with a black hose and black body is hanging on the right side of the device. The hose is corrugated and no visible tears/breaks have been detected. The tip crystal part of the handpiece is not clearly visible in the images.
- External Power Unit: On the left side of the device, a gray metal box on the floor contains an external unit with digital voltage indicators and the “CE” mark. This unit is likely used as a voltage regulator (UPS or regulator). The unit has fan grilles and a power on/off switch.
Quantity Information
The system subject to the report content:
- 1 Diode Laser Hair Removal Main Unit
- 1 Laser Handpiece (integrated/attached to the device)
- 1 Start-up Key (inserted)
- 1 External Power/Regulator Unit (Grey box)
Potential Fault Risk and Evaluation
The device appears physically clean and has powered on without software errors (not displaying error codes). The water flow sensor (Flow: 24) and temperature (22.8) values indicate that the cooling system is operational. The shot count of 165,873 suggests that the handpiece is either at the beginning of its life or lightly used, considering the average life of a diode handpiece (typically 10-20 million shots are promised, but vary in generic devices). However, since diode bar quality varies in such OEM devices, the cleanliness seen in the image does not guarantee technical performance. No mechanical deformation or risk of cable pinching has been observed.