Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 21.04.2026
Report code: 1776781632
Alma Soprano Titanium Laser Hair Removal Device Analysis Report
Device Identification
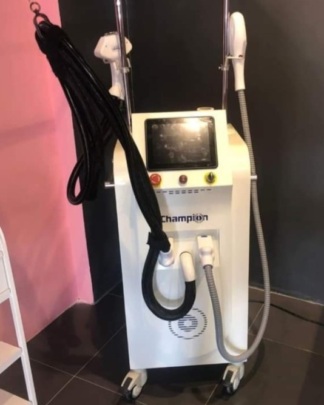
The product in the image is a professional laser hair removal device standing on a wheeled stand, featuring a large screen and two application handpieces.
Brand and Model
The Alma brand is clearly visible on the device body, with the Soprano Titanium model name located directly beneath it.
General Condition and Physical Deformation
The main body, screen, control panel, application handpieces, and wheeled stand of the device are completely wrapped in transparent protective stretch film. This indicates that the device is either new or has been secured for transport/storage. Although fine scratches or details on the surfaces cannot be clearly distinguished due to the stretch film, no visible fractures, cracks, dents, or major physical damage have been detected on the casing, screen, or handpieces. The overall condition of the device appears to be good.
Mechanical and Electronic Components
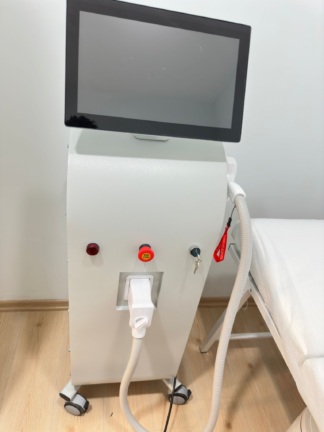
The front panel of the device features one red emergency stop button (emergency switch), one green power button, and a key switch with the key inserted. These control elements appear to be intact and in place. Ventilation grilles are present at the bottom of the device. The wheels of the stand appear to be in good condition. The device screen is off; therefore, no information could be obtained regarding the functionality of the electronic system or the screen menus.
Accessories
Two laser application handpieces are seen attached to their slots on the upper part of the device. These handpieces are connected to the main body of the device via spiral protective hoses. The hoses and connection points appear to be intact. The power cable or other external accessories of the device are not visible in the image.
Label Information and Documents
No labels or plates containing the serial number, lot number, production year, or technical specifications of the device are visible in the image. Furthermore, documents such as invoices, warranty certificates, or user manuals are not present.
Potential Failure Risk
There is no clear evidence of malfunction, such as broken parts or damage, in the images. Given that the device is wrapped in protective film and its physical integrity is maintained, it can be concluded that it is in good condition and does not pose a significant risk of failure.