Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 30.01.2026
MICROSON Aesthetic and Skin Care Device Analysis Report
Device Identification
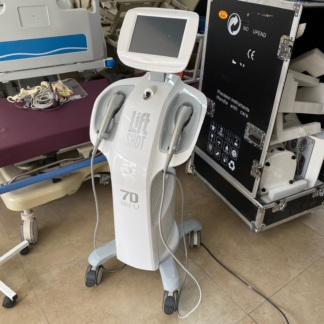
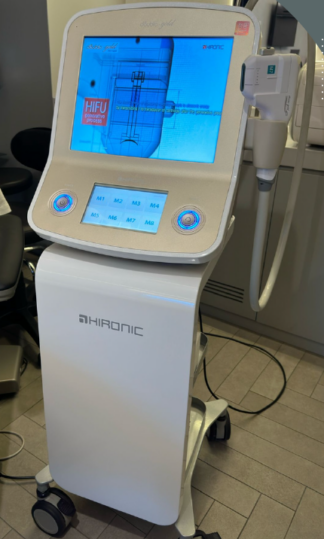
The inspected image shows an aesthetic and medical care device designed in a vertical (tower-type) form, intended for professional use. The device’s main body features an integrated touchscreen panel and a single application head (probe) positioned in the middle section. The design of the device and the structure of its head exhibit characteristics typical of ultrasonic or radiofrequency-based skin treatment systems (usually HIFU systems).
Brand and Model Information
The data obtained from the analysis of the image are as follows:
- Brand: On the front face of the device, directly above the application head’s cradle, the MICROSON logo is clearly and legibly visible.
- Model: No specific text or label indicating a model number (e.g., M-100, Dual, etc.) is visible on the device from this angle. Therefore, the model name is not specified.
Authenticity Assessment
Considering the material quality of the device’s outer casing, the regularity of the brand logo’s printing, the alignment of its parts (screen frame, probe cradle), and its industrial design language, the product is assessed to be an original MICROSON device. No signs of poor craftsmanship typical of aftermarket or counterfeit products (faulty joints, low-quality plastic molding) are observed.
Areas of Use
Considering the MICROSON brand and the structure of the application head (probe) shown in the image, this device is suitable for use in aesthetic clinics, beauty centers, and dermatology offices. Its primary purpose is skin tightening, facial lifting, wrinkle reduction, and body contouring treatments using focused sound waves or similar technologies.
General Condition and Physical State
The cosmetic condition of the device is visually very good and clean. Even if it is in a second-hand status, it gives the impression of being carefully used or well-maintained.
Physical Deformation Examination
- Casing: No cracks, breaks, dents, or deep scratches have been detected on the dark grey/black plastic outer casing. There are no deformations such as color fading or sun damage.
- Surface Cleanliness: No significant dirt, dust accumulation, or stains are visible on the device.
Mechanical and Electronic Components
Based on the visual inspection, the condition of the mechanical and electronic parts is as follows:
- Application Head (Probe): It is fully seated in its cradle. The plastic components of the head’s handle appear intact. The buttons on it are complete.
- Locking Mechanism: The keyed locking mechanism located on the right side of the device is in place, and the keys are attached to it.
- Wheels: A portion of the wheel structure is visible at the bottom, and there is no imbalance in its standing position.
Screen Analysis
A large LCD screen panel is located on the upper part of the device. Since the device is in the off position in the image, the screen is black. However, no impact marks such as cracks, fractures, or dead pixel damage were visually detected on the screen surface. The screen’s matte finish and frame integrity are preserved.
Accessories and Connections
The accessories and equipment identified in the image are as follows:
- 1 Application Head (Handpiece): Grey in color, with a wired connection.
- Key Set: The key used to operate or lock the device is inserted into the ignition slot and is on the keyring.
- Connection Cables: The thick spiral-protected cable and socket input connecting the application head to the device appear intact. No breakage or damage is observed at the cable connection point (strain relief). Power cables are visible hanging in the background, but their ends cannot be clearly distinguished.
Quantity Information
The image shows 1 unit of the main device and 1 unit of an integrated application head.
Potential Malfunction Risk
Based solely on visual data; due to the absence of bending, crushing, or wear on the device’s exterior, particularly at the cable connection points (especially the probe cable’s input socket), the physical potential malfunction risk is assessed as low. It is observed that the device is protected by a “Key” system and the key is inserted, which is a mechanical advantage reducing the risk of unauthorized use.