Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 10.02.2026
Pollogen Regen Body Contouring Device Analysis Report
Device Identification
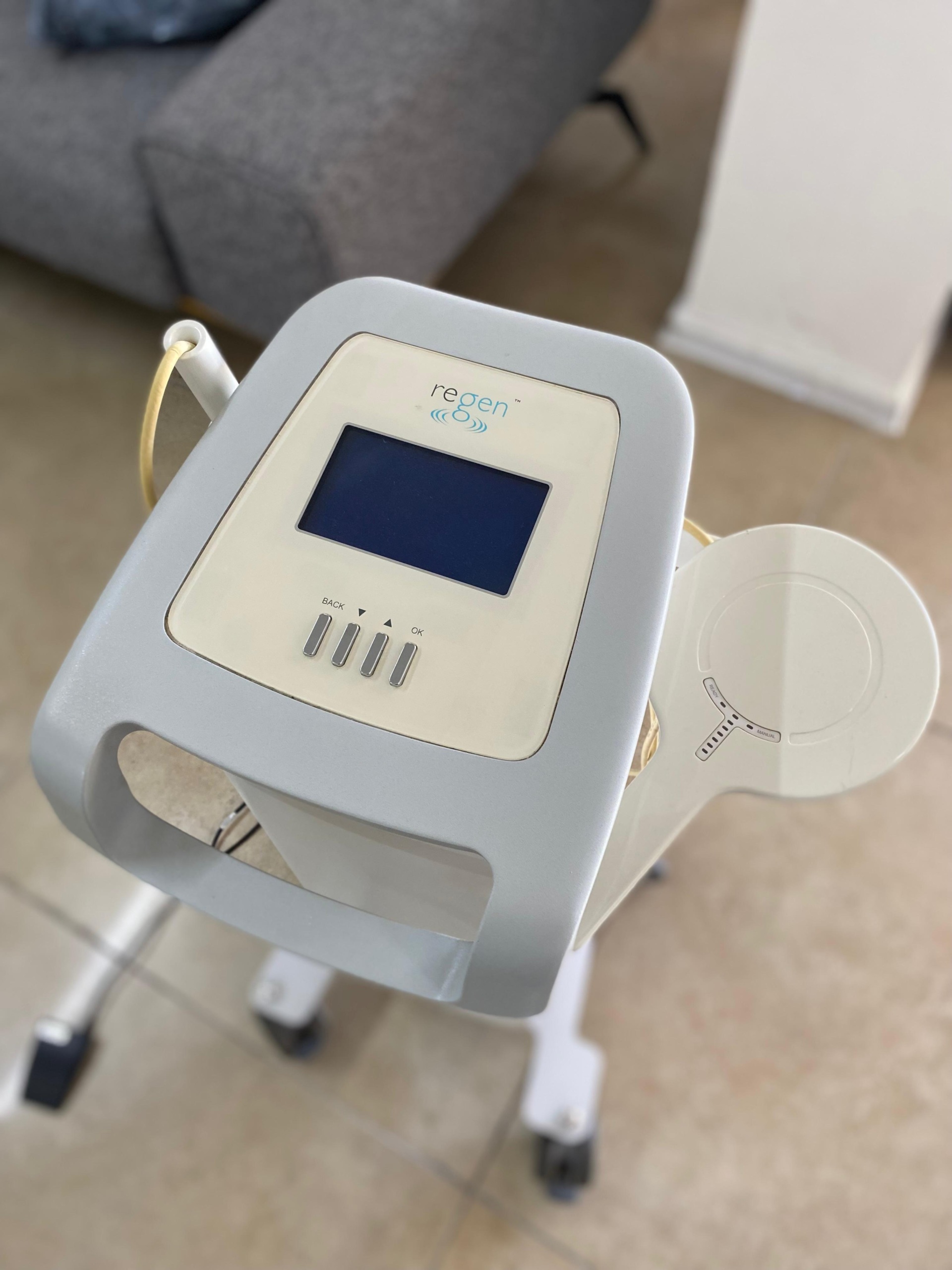
The product in the image is a professional medical aesthetic device designed for aesthetic and medical clinics, featuring a wheeled stand structure. This unit, in a vertical tower form, consists of a single-piece system with a control panel, an integrated handle, and an application head mounted on its side.
Brand and Model
Upon examining the text and logo design on the device’s front control panel, it has been clearly identified that the brand is Pollogen, well-known in the aesthetic device industry, and the model name is Regen. The green-toned logo and the word “regen” located on the device’s top panel confirm this identification. The device carries the characteristic design features of Pollogen Tripollar radio frequency technology.
Originality
The casing mold of the device, the form of the plastic material used, the button arrangement, and the screen placement are consistent with the manufacturer’s original design standards. The probe holder (holster) located on the side and the design of the probe also appear to be original equipment. There is no indication in the image that it is an “aftermarket” or “copy” product.
Areas of Use
Pollogen Regen is a system that utilizes Tripollar Radio Frequency (RF) technology. This device is primarily used for the following purposes:
- Skin tightening and firming.
- Reduction of cellulite appearance.
- Local slimming and body contouring.
- Wrinkle treatments.
It is suitable for beauty centers, aesthetic clinics, and dermatology offices.
Quantity Information
The image shows 1 unit of the main device (stand unit) and 1 unit of an application head (probe) connected to this device via a cable. The device’s power cable is also present in the image.
General Condition
The device is in used (second-hand) condition. Traces of use, accumulated over time, are visible on the product’s outer surface. The device appears to be in a condition of having been actively used in the field and stored, rather than being “new” or “refurbished”.
Physical Deformation
During the physical examination of the product, the following details are noticeable:
- No deep cracks or breaks have been detected on the outer casing, but the surface appears dulled.
- Dirt accumulation and wear are present on the device’s wheels, indicating that the device has been moved on the floor.
- Slight discoloration (yellowing) can be observed on the cable of the application head (probe).
- Slight dirtiness or stains are present on the plastic parts of the front panel.
Mechanical Components
The wheels, which provide the device’s mobility, are in place but may require maintenance. The handle on the front appears sturdy, and no detachment points are visible where it connects to the body. The probe holder mechanism is functional and supports the probe.
Electronic Components and Screen Analysis
An LCD screen panel is located on the upper part of the device. Since the device was turned off during the photo shoot, the screen is black; therefore, pixel errors or image loss cannot be analyzed from the image. There are 4 physical buttons on the panel. From left to right, these buttons presumably represent the functions: “BACK”, Up Arrow, Down Arrow, and “OK”. The markings on the buttons are legible, with no excessive fading observed.
Accessories
The following accessories have been identified on the device:
- Application Head (Probe): A white, large, round-surfaced applicator is hanging on the right side of the device. This is typically the type of head used for body applications.
- Power Cable: A black power cable is wrapped around the device’s handle. Although no distinct breaks are visible on the cable, it is loosely wrapped.
Battery Status
This type of device (clinic-type RF devices) typically operates with mains electricity (AC). There is no indication in the image or in the device’s structure that it operates with an internal battery.
Label Information
The silver label (usually found on the rear side) containing technical specifications (Serial No, Lot No, REF code) is not visible in the front-facing shot of the device. Only the brand/model inscription on the top panel can be read. Therefore, the year of manufacture and technical electrical values could not be determined from the image.
Potential Malfunction Risk
Considering the external condition of the device, the following points are noteworthy:
- Cable Wear: The connection points of the probe cable and the power cable may have become sensitive due to long-term use. Bending points of the probe cable, in particular, should be inspected.
- Need for Cleaning: The wheels and the probe require thorough hygienic cleaning and maintenance.
Apart from these, no damage of a magnitude that would prevent the device from operating is visible on the outer casing; the product maintains its physical integrity.