Ready For Sale
Secondhand Edge HydraFacial MD Tower Skin Care Device
Price: USD$ 15.000,00 Approx: 675.000,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 11.03.2026
Report code: 1773217814
Hydrafacial Skin Care Device Analysis Report
Device Identification and Brand Information
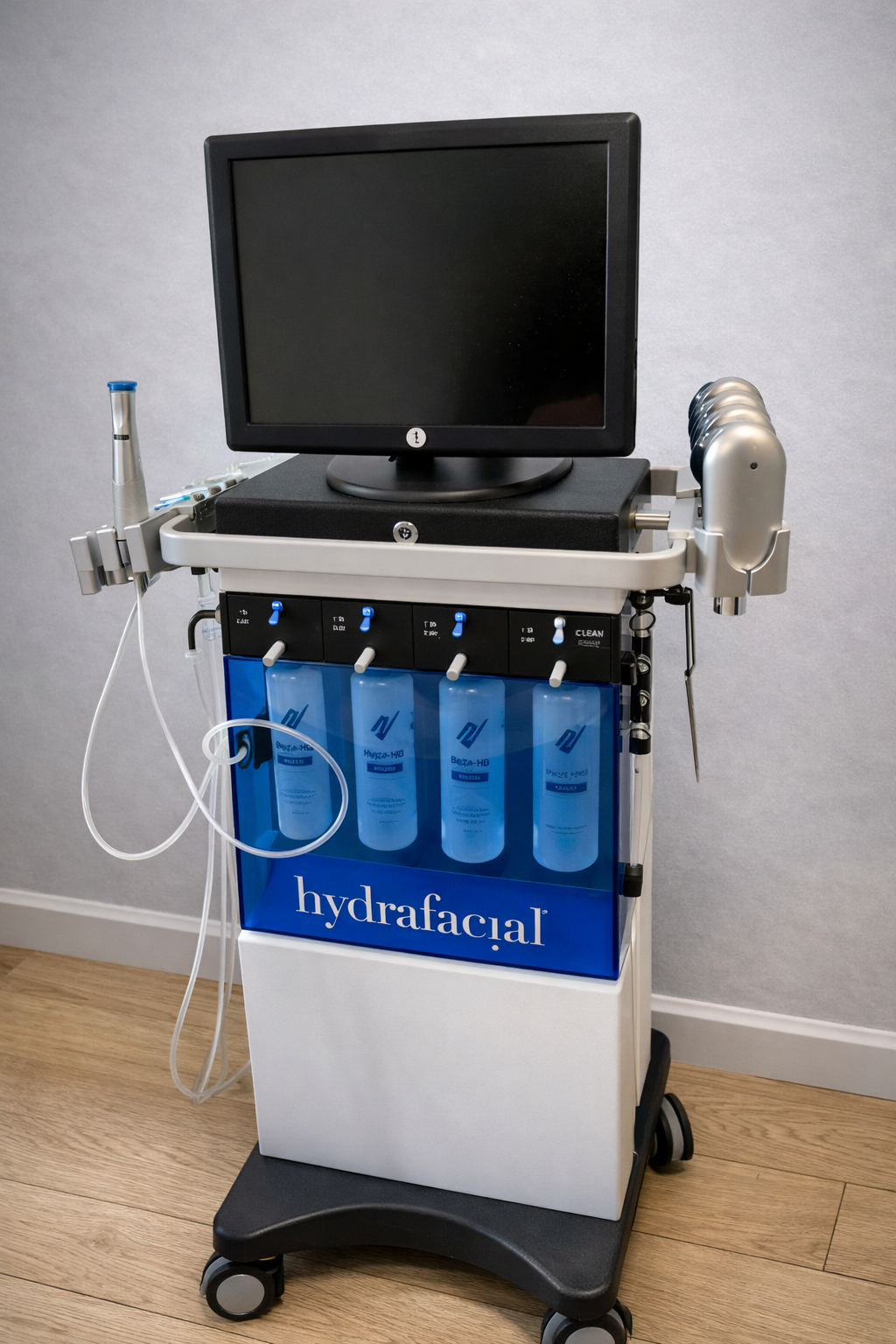
The product in the image is a device used in professional skin care and medical aesthetic applications. Based on the logo written in large white letters on the blue transparent panel located on the front body of the device, its brand has been definitively identified as Hydrafacial.
Applications
This device is used in dermatology clinics, medical aesthetic centers, and beauty salons for professional skin care purposes. The solution bottles and different application tips on it indicate that the device is designed for multi-stage skin care protocols such as skin cleansing, peeling, extraction (pore cleansing), and hydration (moisturizing).
General Condition and Physical State
The general condition of the device is quite clean and well-preserved. No scratches, cracks, dents, or discoloration have been observed on its outer casing, white and black plastic components, blue transparent panel, or monitor. The device appears new or very lightly used, in high condition.
Mechanical and Electronic Components
A black, flat-screen monitor is located on the upper part of the device. The screen is off, and there is no physical damage on its surface. Immediately below the monitor, there is a locked black compartment integrated into the main body of the device. In the middle part of the device, there are four manual valves, understood to control the solution flow, and blue levers belonging to these valves. All levers appear intact. The device stands on a black base unit with four wheels. There is no visible deformation on the wheels.
Accessories and Equipment
According to the count and examination performed via the image, the equipment on the device is as follows:
- Main Body and Monitor: 1 wheeled main stand and 1 top-mounted screen.
- Solution Bottles: 4 solution bottles with blue caps, positioned behind the blue transparent panel.
- Main Application Tip: 1 main application tip (handpiece) with silver and blue color details, resting in the special hanger on the left side of the device. Transparent liquid/vacuum hoses are attached to this tip. There is no crushing or yellowing on the hoses.
- Additional Application Tips: 4 additional application tips, silver-colored and metallic in structure, arranged in the special metal slot on the right side of the device.
Label and Text Analysis
The readable texts and labels on the device are as follows:
- The word hydrafacial is written in capital letters on the front panel.
- On the valve control panel above the solution bottles, the word CLEAN is readable on the rightmost lever. The texts on the other levers cannot be clearly read due to the angle of the image.
- The labels on the bottles contain the Hydrafacial brand emblem and solution names (e.g., Beta-HD).
Potential Malfunction Risk
As a result of the visual inspection, no wear, breakage, rust, or cable crushing has been detected on the device’s casing, screen, hoses, or tips. The physical condition of the device is excellent, and it does not pose any potential malfunction risk based on its external appearance.