Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 15.11.2025
Aeonmed Aeon7400A Anesthesia Machine Analysis Report
Overview and Device Description
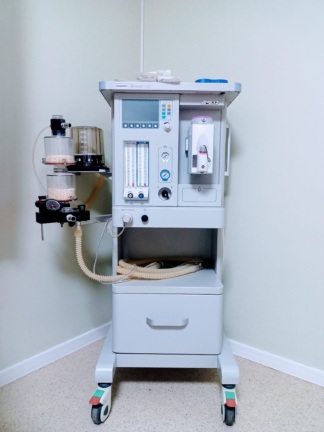
The product shown in the images is an anesthesia machine designed for use in medical operations. The Aeonmed brand and Aeon7400A model information are clearly legible on the product. The device consists of components such as a main control unit, ventilator system, and vaporizer, integrated onto a wheeled stand. The overall build quality and logo print of the product give the impression of an original device. The images show one anesthesia machine and its mounted accessories.
Areas of Use
This device is a system used in hospital units such as operating rooms and emergency departments to administer anesthetic gases and oxygen to patients in a controlled manner. It also supports the patient’s respiratory functions via its ventilator. The device may possess features suitable for use in adult and pediatric patients.
Physical Condition and State
The device generally has a clean and well-maintained appearance, suggesting it is a used but well-preserved product. No significant scratches, dents, cracks, or discoloration have been observed on its metal and plastic components. The overall condition of the device is quite good.
Mechanical and Structural Components
Upon visual inspection of the product’s mechanical components, no issues were detected.
- Carrying Stand: The device is mounted on a metal stand with shelves, two front wheels equipped with locking mechanisms. The stand and wheels appear robust.
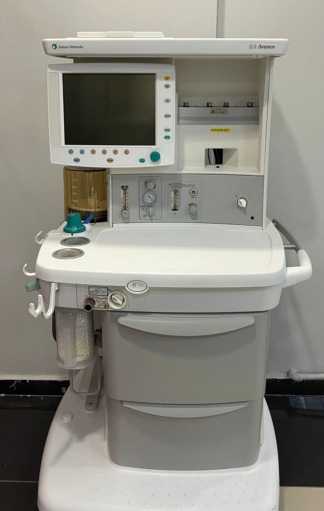
- Control Panel: The pressure gauges (manometers), flowmeters, adjustment knobs, and buttons on the front panel are physically complete and in place.
- Ventilator and Absorber System: The breathing circuit, bellows, and absorber (CO2 absorbent) unit mounted on the left side of the device are structurally sound and free from any breaks or cracks.
- Vaporizer: There is one vaporizer attached to the device, featuring a yellow adjustment knob. The inscription “SEVOFLURANE” is present on this vaporizer. Its mechanism appears functional.
Accessories and Equipment
The images show the presence of some essential accessories required for the device’s operation.
- Blue gas hoses.
- Black and yellow spiral cables/hoses connected to the rear of the device.
- One “SEVOFLURANE” vaporizer.
- Complete ventilator and absorber unit.
Beyond these, the images do not show additional accessories such as an external power cable, user manual, or patient circuit hoses.
Labels and Technical Information
Upon examining the labels on the device, the following information was obtained:
- Brand: Aeonmed
- Model: Aeon7400A
- Vaporizer Gas Type: SEVOFLURANE
- Gas Indicators: OXYGEN and NITROUS OXIDE
There is also a green and white label with “INOVELL” written on it at the top of the device, but other writings on these labels are illegible. Detailed technical information such as the product’s serial number, REF code, lot number, or year of manufacture could not be obtained from the images.
Electronic Components and Display
The front panel of the device features multiple digital displays and LED indicators. However, as the device is not in operation, no comments can be made regarding the functionality of the screens or the presence of any error messages. No physical damage, cracks, or deep scratches are visible on the screen surfaces.
Potential Risk Assessment
Based on visual inspections, no critical deformation, rusting, or cable pinching that would suggest an immediate risk of failure has been detected on the device. The overall physical condition of the device is good. However, such medical devices must undergo calibration and functional tests by an authorized technician before clinical use.