Ready For Sale
Secondhand Ottobock L4D Knee and Hip CPM Device
Price: USD$ 1.730,00 Approx: 77.850,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 12.03.2026
Report code: 1773326200
Otto Bock L4D CPM Device Analysis Report
Device Identification and Application Area
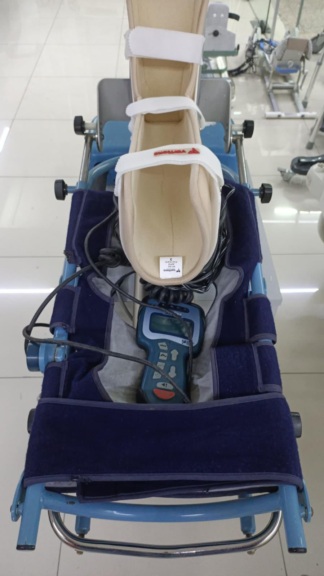
The product shown in the images is a Continuous Passive Motion (CPM) device used in the rehabilitation process of lower extremity (knee and hip) joints after surgery or trauma. It is suitable for use in orthopedics and physical therapy.
Brand and Model Information
Different brand and model information has been identified on the main body and control unit of the device. This situation may stem from the device’s modular structure or manufacturer partnerships.
- Main Body: Otto Bock brand, L4D model.
- Control Unit: QAL Medical brand, OrthoAgility model.
Quantity Information and Accessories
The equipment identified in the images is as follows:
- 1 CPM device main body
- 1 wired hand control unit
- 1 power adapter
- 1 set of grey fleece/plush leg support covers
Originality
Upon examining the device’s metal craftsmanship, plastic molding quality, labeling standards, and the international certification (CE, RoHS) logos on the power adapter, the product is assessed to be an original medical device.
General and Physical Condition
The device is in used condition. No structural defect, bending, or fracture has been observed in the aluminum rail system and the main body. Superficial scratches due to use are present on the plastic motor housings and adjustment knobs. The grey fleece covers on the device are worn out and matted.
Mechanical and Electronic Components
The angle adjustment mechanisms, leg length extension rails, and locking latches are visually complete. No crushing, breakage, or insulation damage has been detected in the unit’s control cable and the power adapter cable. The metal-pinned connectors attached to the main body of the device are intact. The control unit screen is in the off position and shows no physical damage (break, crack). The keypad on the control unit (speed, angle, start/stop buttons) has not been deformed.
Label and Technical Information
The following data has been read from the labels on the device and its components:
- Main Body Label
- Brand: Otto Bock
- Model: L4D
- Serial Number (SN): L4D-00805
- Power Adapter Label
- Model: 3610
- P/N: 2018859-001
- Input: 100-240VAC 50/60Hz 0.5A
- Output: 12VDC 1.0A
- Serial Number (Serial No): 159335
- Manufacturing Date: 06/13
- Place of Manufacture: China
Year of Manufacture
Based on the 06/13 marking on the power adapter’s label, it has been determined that the adapter (and likely the device) was manufactured in 2013.
Potential Malfunction Risk
There is no clear evidence of mechanical or electronic damage in the images that would prevent the device from operating. Cable connections, sockets, and moving parts are in good condition. The overall condition of the device is positive. The fleece covers, which are patient contact surfaces, may need to be replaced due to hygiene standards.