Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 07.10.2025
CardiAid Automated External Defibrillator Analysis Report
Overview and Product Description
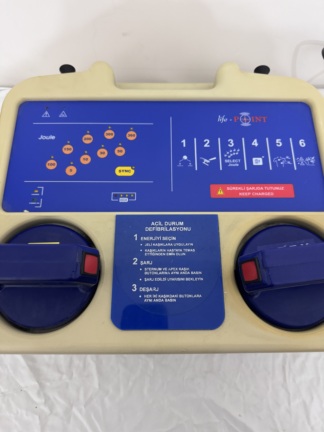
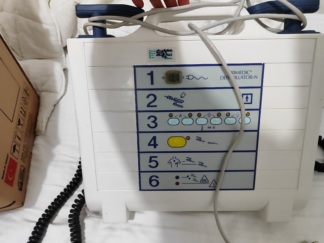
The product shown in the visuals is an automated external defibrillator (AED) device designed for first aid intervention in cases of sudden cardiac arrest. The device has been developed for use by any person with basic life support knowledge in public areas or emergency situations. As understood from the usage instructions in the visual, the device guides the user with audio and visual commands, analyzes the heart rhythm, and administers a life-saving electrical shock when necessary.
The total number of units of the analyzed product is 1 (one) unit. Apart from the device itself and its integrated carrying case, no additional accessories or documents are present in the visual.
Brand and Model Identification
The brand of the product in the visuals is clearly readable as CardiAid on both the outer cover and the inner panel. The model is understood to be an Automated External Defibrillator (AED) designed for public access use. The general name of the device is stated as “CardiAid Public Access Defibrillator”.
Physical Condition and Deformation Analysis
The general condition of the device is very good, and it has a lightly used or near-new appearance. No physical deformation was found during the detailed inspection.
- Casing and Surface: The device’s gray outer casing and orange inner panel are clean. No significant defects such as scratches, dents, cracks, discoloration, or stains are observed on the surfaces.
- Mechanical Components: Mechanical parts such as the carrying handle, lid, and the accessory compartment inside the lid appear visually sound and functional. No broken or missing parts are present.
- Cleanliness: The product is generally free of dirt, dust, or residue. It appears to have been maintained hygienically.
Accessory and Hardware Evaluation
The product is shown with an integrated carrying case. Inside the device’s lid, there is a zippered compartment, likely for storing consumables such as defibrillation pads; however, the contents of this compartment are not shown in the visuals. Therefore, a definitive comment on whether any parts are missing cannot be made. Accessories such as an external charging cable or adapter are not present in the visuals.
Technical Details and Label Information
The device features pictorial (pictogram) instructions showing the usage steps. These instructions explain in simple language how to place the electrodes on the patient and perform cardiopulmonary resuscitation (CPR). However, upon examination of the visuals, specific technical label information such as the product’s serial number, lot number, or REF code is not located in a readable area.
Conclusion and Potential Risk Assessment
In this evaluation based on visual evidence, the CardiAid brand defibrillator device was found to be in very good physical condition. There are no findings on it that would indicate any damage, wear, or malfunction. Its mechanical components are sound, and its overall appearance is clean and well-maintained. The current condition of the device gives the impression that it has been stored correctly and used carefully. Since no serious wear or damage was observed in the visual, no signs of a potential malfunction risk were detected.