Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 29.09.2025
Hewlett Packard 43120A Defibrillator Analysis Report
Overview and Product Description
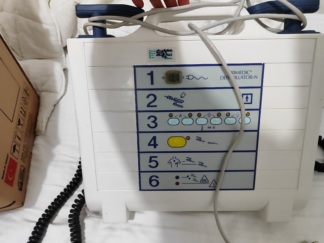
The device shown in the visuals is a Hewlett Packard 43120A model defibrillator. The “hp” logo and “HEWLETT PACKARD” brand information, along with the “43120A DEFIBRILLATOR” model code, are clearly visible on the product. This device is used to apply electrical shocks to the heart and monitor heart rhythm in medical emergencies.
Considering the device’s general appearance, design features, and labels, it is assessed as an original product.
Quantity Information
- Main Unit: 1 unit Hewlett Packard 43120A defibrillator
- Accessory: 1 set (2 units) external defibrillation paddles (pedals) connected to the device with coiled cables
Physical Condition and Deformation Assessment
The device is clearly used. There is a noticeable yellowing on its plastic casing, which has occurred over time and with use. Small scratches and stains were observed in places on its surface, but no cracks, fractures, or major dents were detected.
- Casing: It has a generally robust structure, but its color has turned from the original white/gray tone to yellow.
- Screen: There are no serious scratches or damage on the screen surface. A yellow label with “KONTROL EDİLMİŞTİR” (CHECKED) written on it is present.
- Cable: The coiled cable attached to the defibrillation paddles is physically in one piece, and no significant breaks or crushes are visible. There is dirtiness or aging in the cable’s color.
Mechanical and Electronic Components
The mechanical and electronic components of the device have been visually inspected.
- Mechanical Parts: No visual malfunction or deficiency was detected in mechanical parts such as the “ENERGY SELECT” rotary knob, other buttons, and the slots where the paddles are placed.
- Electronic Components: The device is seen operating in one visual, with power supplied to its screen. The screen displays a straight green line along with the expression “0 P” in the upper right corner. This indicates that the screen and the basic electronic circuit are functional.
Accessories and Connection Points
Besides the integrated external defibrillation paddles (pedals) and their coiled cables visible on the device in the visuals, no additional accessories (power cable, ECG electrodes, battery, etc.) are seen.
Label and Inscription Information
Various informational labels and inscriptions are present on the device. The readable information from these labels is as follows:
- Brand/Model: Hewlett Packard / 43120A Defibrillator
- Usage Instructions: The front face of the device contains instructions in English explaining the usage steps (Charge, Discharge) and recorder operations.
- Warning Labels: A label containing warning information is present on the upper part.
- Control Label: To the left of the screen, a yellow, circular label with “KONTROL EDİLMİŞTİR” (CHECKED) and “BESA” written on it is present.
As a result of examining the visuals, specific production information such as the device’s serial number, lot number, or REF code could not be obtained.
Potential Risk Assessment
The age of the device and the noticeable color change in its casing indicate that it has been used or stored for a long time. This situation suggests that the electronic components, especially the internal battery, may be approaching the end of their operational life. Although the device’s screen is functional, the accuracy of its calibration and defibrillation functions cannot be confirmed by visual inspection alone. It is important that the device be thoroughly checked and calibrated by an authorized technical service before any medical use.