Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 11.07.2025
Nihon Kohden Cardiolife TEC-7621K Defibrillator Product Analysis Report
Basic Product Information
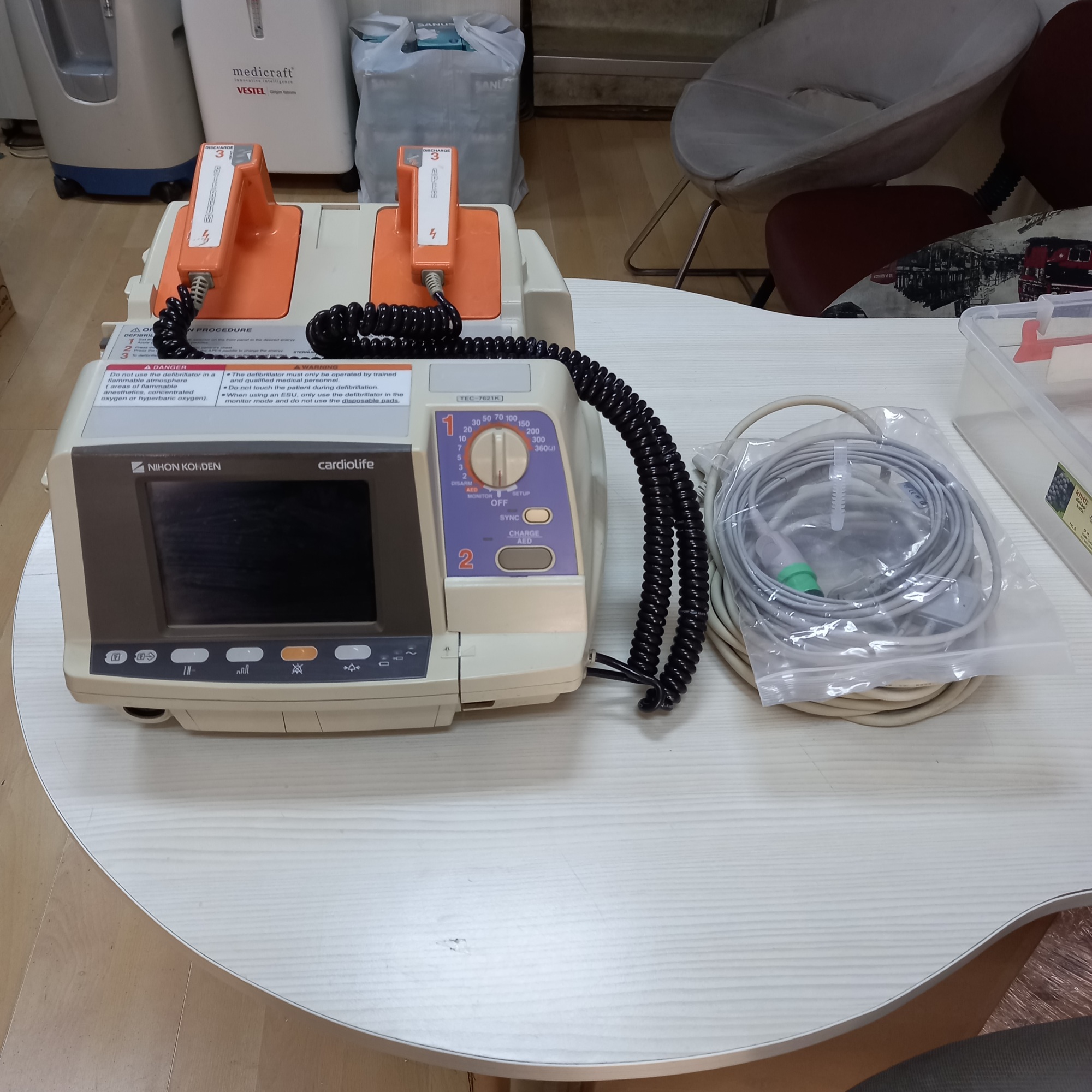
The device in the images is a Nihon Kohden brand, Cardiolife TEC-7621K model defibrillator. The brand and model information are clearly legible on the device. The product is a defibrillator/monitor device designed for professional medical use. Considering the quality of the logo and labels, and the craftsmanship details, the product is assessed to be original.
The device is used to deliver an electrical shock to the heart in emergencies such as cardiac arrest, to restore its normal rhythm. It also functions as a monitor to track the patient’s ECG (electrocardiogram). The images show a total of 1 defibrillator unit and a pair of external shock paddles attached to the device. Additionally, there is an accessory, presumed to be an ECG cable, in a separate bag.
Condition Assessment
The overall condition of the device is assessed as used. There are no significant cracks, fractures, or major deformations observed on its plastic casing. However, there is slight yellowing and superficial scratches on the casing, which can occur over time. This condition does not appear to affect the overall functionality of the device.
- Mechanical Components: The rotary knob used to adjust the energy level and mode, as well as other control buttons, are physically present and appear solid. The orange shock paddles fit properly into their holders. The coiled cable connecting the paddles to the device shows no signs of breakage or crushing.
- Accessories: A pair of shock paddles that come with the device are integrated into it. Additionally, there is a cable with a green connector in a transparent bag. This cable is presumed to be an ECG monitoring cable. No information such as an expiration date was found on the bag or the cable. The power cord or adapter required to charge the device is not present in the images.
- Battery Status: The device’s screen displays a “Charge battery” warning and an empty battery icon. This indicates that the device’s internal battery is depleted or needs to be replaced. No visual swelling or leakage was detected on the battery.
Technical Details and Label Analysis
There are various informational labels on the front panel and top of the device. These labels contain operating instructions and warnings. The label indicating the model number is clearly visible.
- Model Number: TEC-7621K
- Serial Number: A label showing the device’s serial number is not clearly legible in the images.
The device’s screen is in working condition. The information displayed on the screen is as follows:
- Mode and Warnings: At the top of the screen, the “PADDLE” mode and the “Charge battery” warning are visible. There is also an informational message stating “Use disposable pads”.
- Energy Levels: Selectable shock energy levels are indicated as 200J, 300J, 360J. The current shock level is shown as 0J.
- ECG Display: A flat line (asystole) is observed in the area showing the ECG waveform.
Fault and Risk Assessment
The most prominent issue in the images is the “Charge battery” warning on the device’s screen. This is an operational status indicating that the battery needs to be charged or replaced if its life has expired, rather than a malfunction. Since the device’s power cable is not visible in the images, it is unknown whether it can be charged or if a full functional test can be performed.
The slight yellowing and superficial scratches on the device’s casing are cosmetic imperfections and are not considered a potential fault risk. Overall, the physical condition of the device is good. However, as with any medical device, it is recommended that this defibrillator be checked and calibrated by a qualified technical service to ensure it operates at full performance.
Note: The potential risk assessments in this section are based solely on visual inspection and do not mean that a fault definitively exists or will occur.