Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 30.12.2025
PHYSIO CONTROL LIFEPAK 1000 Defibrillator Analysis Report
Device Identification
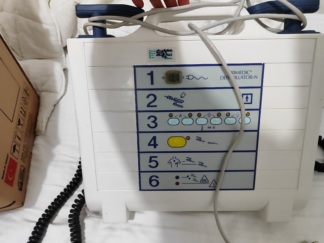
The device shown in the visuals is a high-tech defibrillator intended for professional use. Specifically, it is a portable medical equipment designed to intervene in life-threatening cardiac conditions, falling into the category of Automatic External Defibrillators (AED).
Brand and Model
As a result of detailed visual inspection, the brand and model information of the device have been definitively identified:
- Brand: PHYSIO CONTROL
- Model: LIFEPAK 1000
In the visuals, the inscription “LIFEPAK 1000 DEFIBRILLATOR” in white font is clearly visible on the front upper part of the device, and the “PHYSIO CONTROL” logo within a square is visible on the lower part. This information has been verified by an internet search.
Originality
The quality of the logos on the product, the professional printing of warning labels (especially the orange warning film on the screen), the “QUIK-COMBO” branded electrode sets, and the original carrying case indicate that the product is definitively an original PHYSIO CONTROL production. There is no indication of it being an aftermarket or replica product.
Areas of Use
The LIFEPAK 1000 is used to restore normal heart rhythm (defibrillation) in patients experiencing sudden cardiac arrest. This device is particularly suitable for the following areas:
- Ambulances and Emergency Medical Technicians (EMT/Paramedics)
- Hospital emergency departments and intensive care units
- Fire and search and rescue teams
- Occupational health and safety units
Quantity Information
The total product content identified in the visuals is as follows:
- 1 Unit LIFEPAK 1000 Defibrillator Device
- 1 Unit Black Original Carrying Case (Not a hard case, but a soft protective cover/bag)
- 1 Pack QUIK-COMBO REDI-PAK Defibrillation/ECG Electrode (Adult compatible)
- 1 Unit Lithium Battery (Inside pink anti-static package)
- 1 Unit yellow-tipped data/test socket-like part (Attached in the upper left slot of the device)
General Condition and State
The device appears to be in unused (new) or factory condition. The strongest evidence for this is the orange protective film on the device’s LCD screen, which states in English “Remove this label prior to placing the device in use.” This film has not yet been removed. There are no signs of use on the keypads, casing, or connection points.
Physical Deformation
No scratches, breaks, cracks, discoloration, or impact marks have been observed on the device casing (yellow and black plastic areas). The device is cosmetically in 10/10 condition. The stitching and structure of the carrying case appear intact.
Mechanical and Electronic Components
The “ON” (Power On/Off), Shock (Lightning icon), and Menu buttons on the device are intact and in place. The “Key” and “Battery” symbols visible in the upper right corner of the screen indicate that the LCD screen is physically sound. However, as the device’s powered-on state was not displayed, its electronic functionality has not been tested.
Accessories and Dates
The condition of the critical accessories included with the device is as follows:
- Electrodes (Pads): “QUIK-COMBO REDI-PAK” packaged pads are present in the inner mesh compartment of the bag lid. The package is closed and sealed. The word “ADULT” in white on a red background is visible on the package.
- Battery: An unopened battery is present in a pink anti-static bag. Label details are legible in the visual.
Label Information
The data read from the label on the battery package is as follows:
- Part Number (Cat #): 11141-000156
- Lot Number (LOT): 1742
- MIN Code: 3205379-006
- Expiration Date (USE BY): 2023-10-17
Usage Duration
Since the protective label on the screen is still present, it is assessed that the device has not been actively used in the field, meaning its usage hours are likely 0 or solely for testing purposes.
Potential Malfunction Risk and Critical Warning
There are no signs of physical malfunction in the visuals. However, there is a very important point to note in the report:
Battery Has Expired: The expiration date on the battery package is October 17, 2023. As of the report date (2025), this battery has expired. The risk of lithium-based medical batteries losing capacity or not being accepted by the device when past their expiration date is very high. A new and in-date battery must be procured for the device to operate. Similarly, although the expiration dates of the electrode pads cannot be read from the visual, if they were procured at the same time as the battery, their expiration date is also likely to have passed. The buyer should replace these consumables.