Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 13.12.2025
Nihon Kohden Cardiofax ECG-9620L Electrocardiograph Device Analysis Report
Device Identification and Areas of Use
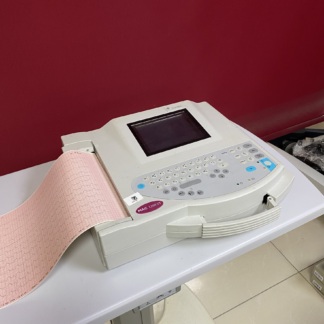
The product in the visuals is an electrocardiograph (ECG) device, which is a medical diagnostic device. This device is used in cardiology departments of hospitals, emergency services, and outpatient clinics to record the heart’s electrical activity and report it graphically. The product consists of a main unit and patient cables connected to it. It is understood to be a medical professional device.
Brand and Model
The brand is clearly readable on the upper left corner of the device’s front surface, the series name on the control panel, and detailed model information on the bottom label.
- Brand: NIHON KOHDEN
- Series Name: Cardiofax
- Model: ECG-9620L
Originality
Upon examining the processing of the logo on the product, the typography of the labels, the material quality, and the certification information on the back cover (CE mark and brand registrations), the product is assessed to be an original Nihon Kohden production.
General Condition and Physical Deformations
The device appears to be in “used” condition upon visual inspection. There are traces of use accumulated over the years.
- Color Change: In the device’s gray-white plastic casing, slight yellowing and tonal differences, accumulated over time, are noticeable, especially on the top and side surfaces.
- Dirtiness: Accumulated dust and dirt are present between the device’s buttons, in the ventilation grilles, and on its surface.
- Casing Integrity: No noticeable deep cracks, breaks, or dents have been detected on the casing. The keypad’s coating appears intact, but there are superficial stains due to use.
- Feet: The rubber feet on the bottom of the device are in place.
Technical and Electronic Components
Electronic components visible from the device’s external casing have been examined:
- Screen: The LCD screen located in the upper right section is off. No physical cracks or fractures are visible on the screen.
- Control Panel: The keypad (membrane buttons) is physically intact. Function keys like F1, F2, F3, and their symbols are readable.
- Printer Unit: A built-in thermal printer unit is located on the left side of the device. The printer cover is closed.
- Ports: The side panel features two audio/signal input-output ports, one serial port (RS-232 like), and a grounding screw. The physical condition of the ports appears good.
- Power Input: The standard IEC power input socket is intact.
- Internal Circuits: When looking through the ventilation grilles at the bottom, internal cables (red, yellow, black, etc.) are discernible, indicating that the device’s interior is not empty.
Accessories and Quantity Information
The following parts, including the main device, have been identified in the visuals:
- 1 Unit NIHON KOHDEN ECG-9620L main unit.
- 1 Power cable (Schuko type plug).
- 1 bundle of ECG patient cables, lying mixed in a transparent bag. (A precise number cannot be provided as the cable ends and quantity are mixed, but they are data cables compatible with the device).
- 2 blue extremity (clip) electrodes.
The cables are seen to be scattered and tangled in the bag. The ECG clips are also in used condition.
Label Information
The following technical details have been obtained from the main label located on the bottom surface of the device:
- Model: ELECTROCARDIOGRAPH MODEL ECG-9620L
- Power Input: 220V, 50/60 Hz, 45VA
- Serial Number (SN): 01724
- Manufacturing Year: 2004
- Manufacturer: NIHON KOHDEN CORPORATION (Although “Made in Japan” is not visible, the brand’s origin and label language point in this direction; a Tokyo address is specified.)
- Representative: NIHON KOHDEN EUROPE GmbH (Germany address)
- Compliance: CE 0086 Certification mark is present.
Battery Status
The label includes battery information: “Battery Pack SB-901D (12V / 1950mAh)”. This information indicates that the device has a rechargeable battery slot for portable use. However, as the battery cover has not been opened, it is unknown whether a battery is inside or, if so, its condition (swollen, depleted, etc.).
Manufacturing Year
The manufacturing date is clearly stated as 2004 on the label at the bottom of the device.
Dimensions and Compatibility
The device is of compact (portable) dimensions. While patient compatibility depends on the electrodes used and software settings, the control panel in the visual features both adult and child figures, indicating that the device has modes suitable for use with pediatric and adult patients.
Current and Potential Risk Analysis
No physical breakage or missing parts (casing-wise) are visible on the device. However, being manufactured in 2004, the device may have largely completed its technological lifespan. As the device is powered off, there is no information regarding its electronic functionality or screen pixel status. The mixed and crushed appearance of the accessories (cables) may pose a risk of poor contact within the cables.