Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 14.02.2026
Biomedical Instruments DMS 9800+ Holter Recorder Analysis Report
Device Identification
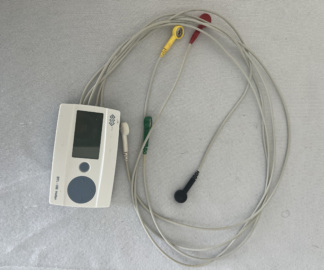
The device shown in the visuals is a Holter ECG Recorder, which is a medical diagnostic and monitoring equipment. The device is a portable unit that can be carried by the patient, used to record heart rhythm over an extended period (typically 24-48 hours or longer). It comes with a carrying case, electrode cables, and a user manual.
Brand and Model
The device in the visuals has been thoroughly examined in light of the data on the user manual and packaging. The phrase “DMS 9800 + HOLTER RECORDER” written in large print on the cover of the user manual ensures the precise determination of the model. Furthermore, the expression “BI Biomedical Instruments for Metko” and its logo are located at the bottom of the manual. Although the device’s front panel displays “Holter ECG,” a general description instead of the model name, the device information based on matching documents is as follows:
- Brand: Biomedical Instruments (As stated in the manual, manufactured for Metko)
- Model: DMS 9800+ (or DMS 9800 Plus)
Originality Assessment
The original printed, spiral-bound “Operator Manual” (User Manual) included with the product, the carrying case with a custom-cut foam insert perfectly matching the device’s form, and the craftsmanship of the keypad and screen on the device indicate that the product is an original set. Branded documentation and professional packaging, not typically found in aftermarket or counterfeit products, are present.
Areas of Use
This device is designed for use in Cardiology and Internal Medicine specialties. Its primary purpose is to continuously record heart rhythms during the patient’s daily life (Ambulatory ECG monitoring). It is used in situations such as the detection of rhythm disorders (arrhythmia), investigation of causes of fainting (syncope), and control of pacemaker functions. The device is a diagnostic tool.
Quantity Information
The set shown in the visuals consists of 5 main parts in total:
- 1 Holter Recording Device (Main unit)
- 1 Spiral-Bound User Manual
- 1 Plastic Carrying Case (Foam-padded)
- 1 Patient Cable Set (Multi-cable, in a bag)
- 1 AA Battery
General Condition
The general condition of the product is very good. The device and its accessories appear to have been carefully preserved. The internal foam padding of the carrying case is clean and undeformed, proving that the device has been well maintained. The overall appearance of the set suggests that the product has either been used very little or stored without use (the way the cables are coiled supports this).
Physical Deformation
No breaks, cracks, or dents have been detected on the device’s outer casing, corners, or connection points. The color of the yellow buttons on the keypad is vibrant, with no signs of fading or wear. There are no deep scratches on the black plastic casing.
Mechanical Components
The locking mechanisms (blue clips) and hinges of the carrying case appear sturdy. The buttons on the front panel of the device (directional keys and enter key) are in place. The connection port at the bottom of the device appears mechanically sound, with no visible bending of the pins.
Electronic Components
There is no visual evidence as to whether the device is operational (as there is no image showing the screen turned on). However, the externally visible electronic ports of the device (the multi-pin input at the bottom) are clean. No signs of oxidation or corrosion are observed.
Accessories and Connection Points
Patient Cables: The set includes a multi-lead ECG patient cable found within a transparent bag. The colored sockets on the cables (red, yellow, green, etc.) indicate standard ECG derivations. The neat coiling of the cables and the cleanliness of the connector end are noteworthy.
Connection Port: At the bottom of the device, there is a large, multi-pin (likely used for both data transfer and patient cable connection) special socket input. This input appears undamaged.
Battery Status
Next to the device, there is 1 VARTA Industrial Pro brand AA type battery. It is understood that the device’s power source is provided by a replaceable battery. No leakage or deformation is visible on the battery. The charge level of the battery cannot be determined from the visual.
Label Information
Important technical and version information located on the cover of the user manual is as follows:
- Document Name: DMS 9800 + HOLTER RECORDER Operator Manual
- Version: Version 3.2
- Document Date: Date: Oct 2013
- Document No.: Document No.: BI/TFCE-11-10-EN
- Company Information: Biomedical Instruments for Metko
Screen Analysis
The device features a small LCD screen, presumed to be monochrome (single-color). Since the device is off, no error codes, menus, or warnings are visible on the screen. However, upon careful examination of visual 3, slight superficial marks or reflections are noticeable on the screen glass (or its plastic protector), but there are no cracks that compromise the screen’s integrity.
Size and Compatibility
The device has a compact, palm-sized design (its size can be understood when compared to an AA battery). It is of a general-purpose size for adult and pediatric use (when appropriate electrodes are used).
Year of Manufacture
The exact manufacturing date of the device is not readable from the label, but the publication date of the user manual included with the device is stated as October 2013 (Oct 2013). This indicates that the device might have been manufactured around or shortly after this date (between 2013-2015).
Documents
Only the “Operator Manual” (User Manual) is included in the set. No invoice or warranty certificate is present in the visuals. Considering the 2013 document, it is confirmed that the product’s factory warranty has expired.
Period of Use
There is no external counter on the device indicating total operating hours. Software data could not be accessed because the screen was off.
Existing Faults
As a result of visual analysis, no existing faults that compromise the physical integrity of the device (such as breaks, severed cables, or dented ports) have been detected.
Potential Fault Risk
The cleanliness and preservation of the device are noticeable in the visuals; therefore, there is no visible, acute risk. However, the device having documentation from 2013 indicates that it is a technology over approximately 10 years old. The aging of electronic components in devices stored unused for a long time is normal, but visually, its condition is at a “reassuring” level.