Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 25.10.2025
Analysis Report of EMS Sienna EEG Device and Computer Set
Overview and Device Identification
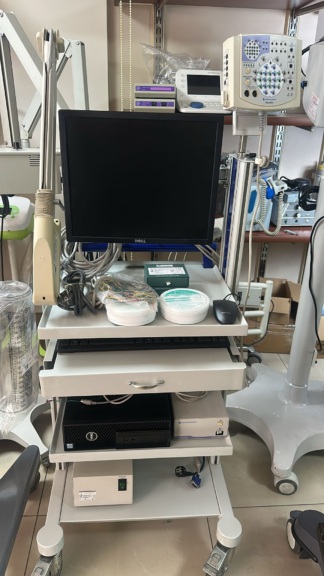
The visuals display a complete medical diagnostic system and its components, used for neurological diagnosis. The system consists of an EEG (Electroencephalography) amplifier unit, numerous electrodes connected to this unit, a computer set (monitor, case, keyboard, mouse) used to control the system and display data, a printer, a camera, and a wheeled medical trolley that houses all this equipment. The electrode input labels on the device (Fp1, Fz, A1, etc.) and its general structure indicate that it is used for neurophysiological measurements such as recording brain electrical activity. The set also includes two boxes of “Ten20” branded conductive paste.
Brand and Model Information
As a result of the detailed visual inspection, the brand and model information of the system’s components has been identified as follows:
- EEG Device: The EMS brand and Sienna model name are clearly visible on the device.
- Medical Trolley: The tupmed brand is visible on the side of the white and wheeled shelf where the system is mounted.
- Monitor: It is LG branded.
- Printer: It is Brother branded.
- Mouse: It belongs to the Logitech brand.
- EEG Paste: The Ten20 brand is found on the boxes.
The brands of the computer case, keyboard, and the camera on the monitor cannot be identified from the visuals.
General Condition and Physical Deformations
The system is shown set up and in working condition, suggesting it is in use at a healthcare institution or laboratory. Generally, it is observed to be in a lightly used and well-maintained condition. There are no significant deformations such as prominent scratches, cracks, dents, or discoloration on the device casings, monitor screen, or medical trolley. Although the numerous cables forming the electrodes have a tangled appearance due to their structure, no signs of crushing, breakage, or wear are observed in the cables. All components have a minimum level of dust that can occur with normal use.
System Components and Accessories
The system in the visuals is a complete set, combining the main components necessary for its operation and some consumables.
- EMS Sienna Amplifier (Headbox): This is the main recording unit to which numerous colored electrode cables are connected. It has markings indicating scalp positions (Fp1, Fp2, Fz, Cz, etc.) used for standard EEG recordings.
- Electrode Set: A complete set of electrodes, consisting of cables in various colors (yellow, green, red, purple, etc.), is connected to the amplifier.
- Computer Set: It consists of an LG branded monitor, a PC case with red and black details, a black colored keyboard, and a Logitech branded optical mouse. The monitor screen is on, and a login screen displaying the username “MEDELEK” is active.
- Other Accessories: The set includes a Brother branded printer, a webcam placed on top of the monitor, and a tupmed branded wheeled trolley stand that makes the system mobile. No main component appears to be missing.
Labels and Technical Information
The following information can be partially read on the labels located on the EMS Sienna EEG amplifier:
- Model: Sienna
- Code: ID 2200A 01… (rest is not clear)
Apart from this, labels providing detailed technical information such as serial number or year of manufacture cannot be discerned in the visuals.
Potential Failure Risk
Based on the inspections in the visuals, no signs of a failure requiring immediate intervention or serious damage have been detected in any part of the system. No negative findings regarding cable and connector wear, which is the most common problem in such devices, have been encountered. The generally clean and well-maintained condition of the devices suggests regular use and care. Therefore, according to visual evidence, the risk of the system experiencing a failure in the near future is assessed as low.