Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 27.11.2024
Celon CelonLab ENT Medical Radiofrequency Device and Set Analysis Report
Device Identification
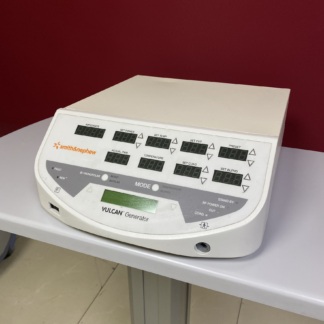
The product in the image is a radiofrequency ablation and coagulation system used in ENT (Ear, Nose, Throat) surgical procedures. The set consists of its box, which includes the main unit (generator), foot pedal, application electrodes, connection cables, and a transport/starter set.
Brand and Model
As a result of a detailed visual inspection, the brand and model information of the product have been identified as follows:
- Brand: CELON (Stated as “Celon AG” in the user manual.)
- Main Device Model: CelonLab ENT
- Box/Set Model: Celon ENT – Starter-Set
- Product Component Models (Stated on the box): CelonProBreath, CelonProSleep, CelonProSpeech
Originality
The overall structure of the product, the consistency of the logos on it, the original “Celon AG” printed user manual, and the packaging’s compliance with medical device standards indicate that the product is an original Celon production.
Areas of Use
As can be understood from the “ENT” (Ear, Nose, Throat) inscription on the device and the intraoral/palate drawings seen in the manual, this device is used in Ear, Nose, and Throat operations. The inscriptions “ProSleep” (sleep apnea/snoring treatment), “ProBreath” (breathing problems), and “ProSpeech” written on the box indicate that the device provides treatment by applying radiofrequency to tissues such as the soft palate and nasal turbinates.
Quantity Information
The count of parts presented as a set in the image is as follows:
- 1 unit CelonLab ENT Main Unit
- 1 unit “Celon ENT – Starter-Set” Cardboard Box
- 1 unit Blue Colored Foot Pedal (inside a nylon bag)
- 1 unit User Manual / Booklet
- 2 units Probe/electrode in sterile packaging (packaged)
- 3 units Unpackaged connection cable/probe (with different tip structures)
- 1 unit Electrical Power Cable (black cable group attached to the device or beside it)
General Condition
The set generally appears used but well-maintained. The main unit looks clean. While some of the consumables (the packaged ones) appear unused, the condition of the exposed cables is visually good.
Physical Deformity
- Box: The “Celon ENT – Starter-Set” box has noticeable wear, cardboard deformations, and discoloration on its corners and edges. It is observed that the box lid is bent, giving the impression that it has been stored on shelves for a long time or transported.
- Main Unit: No breaks, dents, or heavy scratches have been detected on the casing, visible front panel, or metal/plastic parts of the device. It is cosmetically clean.
Mechanical Components
While a clear comment cannot be made about the mechanical condition of the foot pedal as it is kept in a transparent bag, it is estimated to be in good condition due to its protected state. No visible breaks or abrasions have been noticed in the outer insulation of the cables.
Electronic Components
The front panel, indicators, and power input of the main unit appear sound. However, since the device’s screen or indicator lights are not displayed in the “power on” position (emitting active light), a definitive judgment about its electronic functionality cannot be made from the visual.
Accessories
The set has a rich accessory content:
- Different-tipped cables, understood to be probes, are present inside and next to the box.
- A foot pedal is present and in its bag.
- The packaging integrity of the products in sterile packages appears intact in the visual, but the sterility status should be closely inspected.
- The manual contains visually illustrated diagrams.
Label Information
The readable technical codes on the box and packaging are as follows:
- REF Code (On Box): SET990.003.007
- Box Content Description: Bipolar Coagulation Electrodes for ENT
- Product Names (On Box): CelonProBreath, CelonProSleep, CelonProSpeech
Documents
Along with the product, there is an illustrated user manual with the Celon AG logo, showing the device’s areas of use and application points. Invoice or warranty documentation is not present in the visual.
Potential Malfunction Risk
Although the product is in good visual condition, there might be wear or oxidation at the tips of the consumable probes (cables) due to use, but these details are not clear in the distant photograph. The expiration dates of the sterile packages cannot be read from the visual; expired sterile products pose a medical risk and cannot be used. Therefore, the dates on the packaging must be checked. The severe wear on the outside of the box suggests that the device might have been stored in a warehouse for a long time.