Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 10.05.2026
Report code: 1778410541
Nihon Kohden Neuropack S1 EMG/EP Device Analysis Report
Device Identification
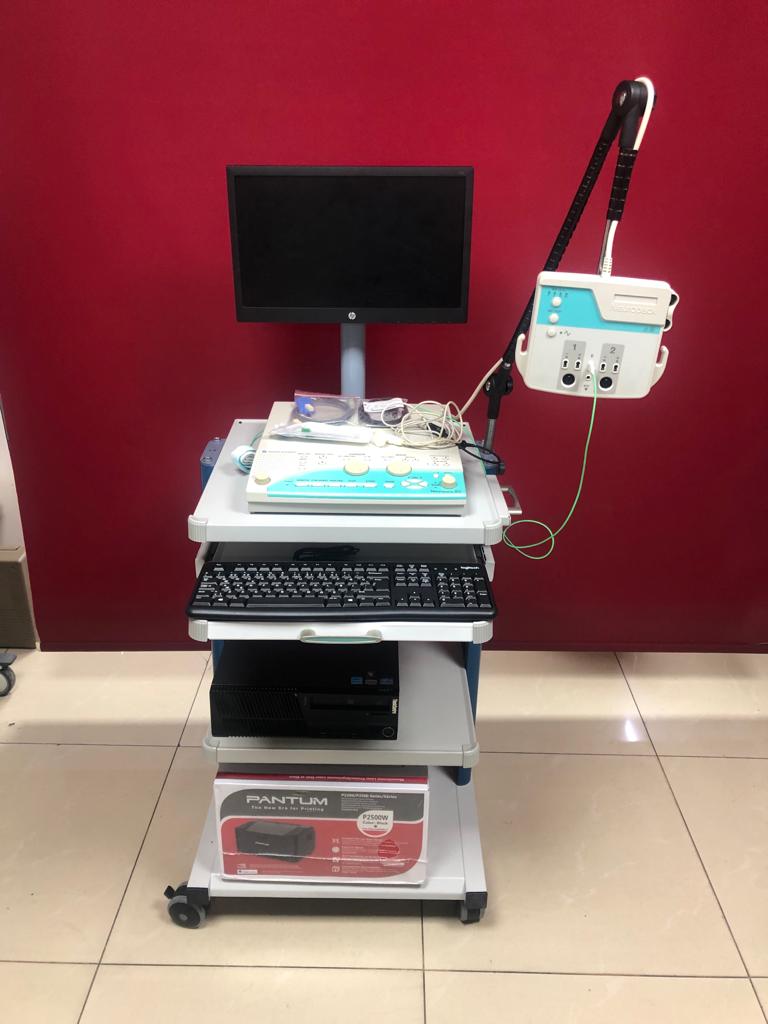
The product in the images is an electromyography (EMG) and evoked potential (EP) measurement device used for medical purposes. The system consists of a main control unit, a patient input box, a monitor, a computer tower, a keyboard, and various connection cables and accessories. All components are mounted on a wheeled transport cart.
Brand and Model
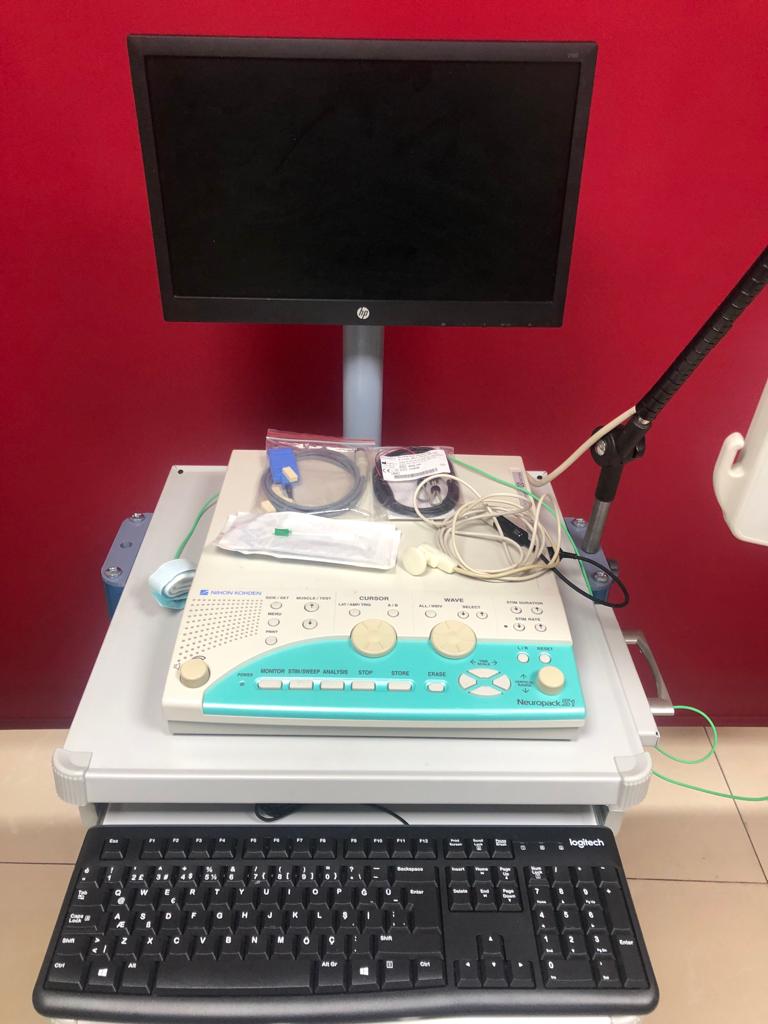
Detailed examination of the main control unit and the patient input box clearly identifies the brand and model information. The Nihon Kohden logo is visible on the top left corner of the main unit, and the Neuropack S1 model name is legible on the bottom right. The Neuropack label is also present on the patient input box. The monitor used in the system is an HP brand, and the keyboard is a Logitech brand. The brand of the computer tower cannot be clearly identified from the images.
Authenticity
The quality of the logos on the device, the placement of the labels, the design of the control panel, and the overall material quality indicate that the product is original. The control buttons and socket inputs appear to comply with standards.
Usage Areas
This device is used in fields such as neurology, physical medicine, and rehabilitation to evaluate nerve and muscle functions. It allows for nerve conduction studies, needle EMG, and various evoked potential tests. It plays a critical role in the diagnosis of muscle diseases, nerve entrapments, and other neuromuscular disorders.
Quantity Information
There is one complete EMG/EP system set in the images. This set includes 1 main control unit, 1 patient input box, 1 monitor, 1 computer tower, 1 keyboard, and 1 transport cart. Additionally, various connection cables and electrodes are visible on the main unit, both in bags and loose.
General Condition
The general condition of the device appears used but clean and well-maintained. All parts are together and assembled. Its placement on the transport cart is organized.
Physical Deformation
No noticeable deep scratches, cracks, fractures, or color fading have been detected on the plastic casing, control panel, or monitor of the device. The text on the keypad is legible. It has a generally clean appearance.
Mechanical Components
The wheels of the transport cart, the shelves, and the movable arm holding the patient input box appear sturdy. The rotary knobs and push buttons on the main unit are in place. No mechanical issues have been observed.
Electronic Components
The internal electronic components of the device are not visible. External inspection shows that the socket inputs are clean and intact. Cable connections are in place. There is no image on the monitor to indicate whether the device is operational.
Accessories
Various connection cables, electrodes, and one grounding strap are visible on the main unit, both in bags and loose. There is a green cable connected to the patient input box. The expiration dates of the accessories are not readable from the images. There are 2 channel inputs and reference/ground inputs on the patient input box.
Battery Status
There is no information in the images regarding whether the device has an internal battery. The system operates by being connected to the mains via the computer tower.
Label Information
Technical labels on the back or bottom of the device are not present in the images. Therefore, specific information such as serial number, lot number, or year of manufacture could not be determined. Only the model name and brand logo on the main unit are available.
Monitor Analysis
The HP brand monitor used in the system is turned off. Therefore, no software interface, error message, or patient data is visible on the screen.
Dimensions and Compatibility
There is no clear measurement information regarding the physical dimensions of the device. However, its stance on the wheeled cart indicates that it is of standard medical device size. The sockets on the patient input box are compatible with standard EMG/EP electrodes.
Documents
No invoices, warranty certificates, or user manuals belonging to the device are visible in the images.
Usage Duration
There is no information on the screen or labels regarding the total operating hours or duration of use of the device.
Existing Faults
There is no clear evidence in the images of the device being faulty, such as a broken part, severed cable, or physical damage. Since the device is turned off, it cannot be determined whether there is an electrical fault.
Potential Fault Risk
The physical condition of the device is good. No crushing in the cables, corrosion in the sockets, or significant wear on the casing has been observed. Therefore, based on the visual data, it can be said that the potential fault risk of the device is low and it is in good condition.