Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 31.07.2025
Viasys Healthcare Medelec Synergy EMG Device Analysis Report
Product Overview and Content
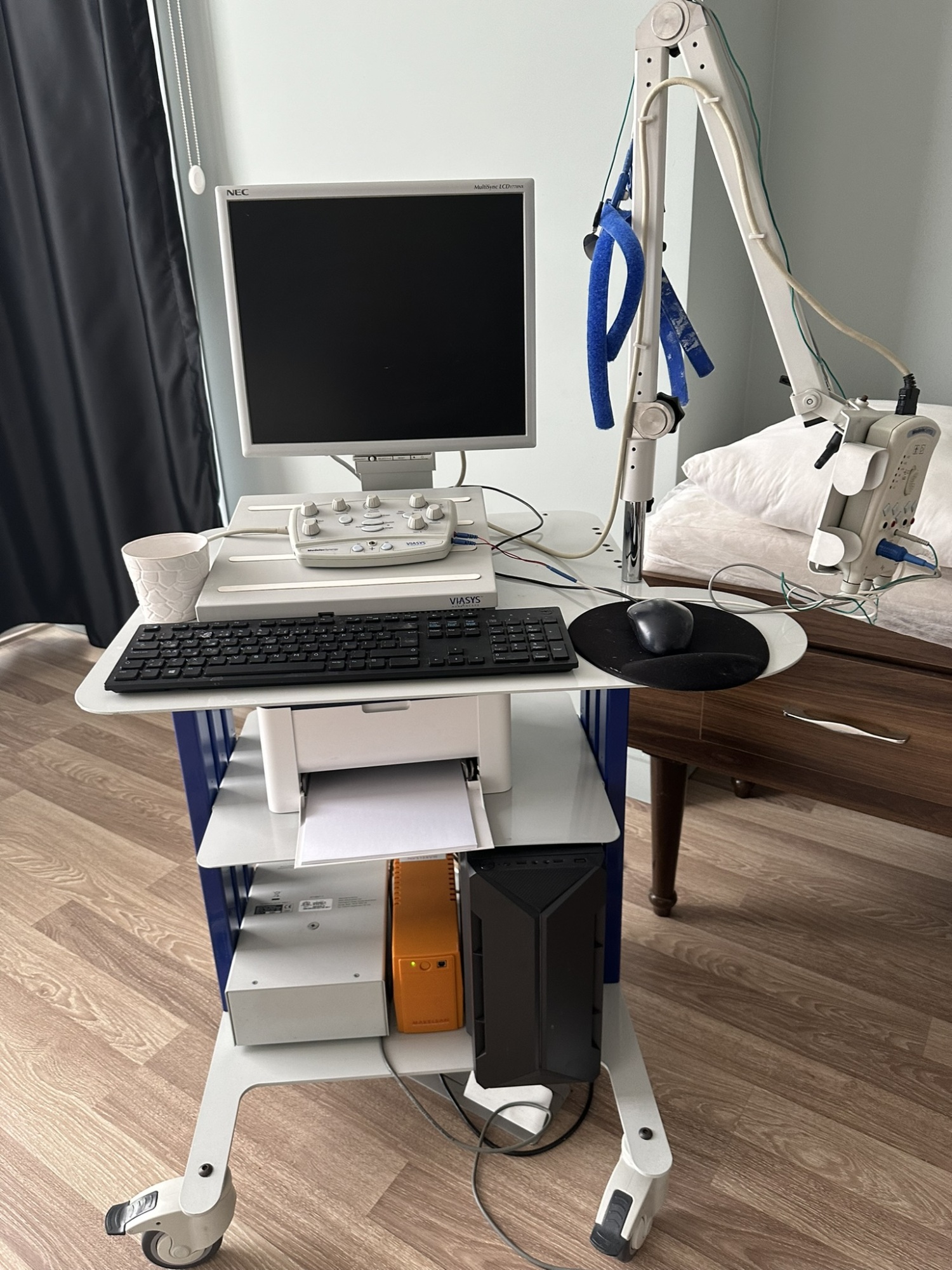
The images show a multi-component medical diagnostic system mounted on a wheeled stand. The labels and markings on the product indicate that it is a Viasys Healthcare Medelec Synergy electromyography (EMG) and nerve conduction studies (NCS) system. The system consists of a computer, monitor, control units, an amplifier, and various peripherals.
The products in the image are as follows:
- 1 main carrier stand with wheels and an articulated arm
- 1 Viasys branded main control unit
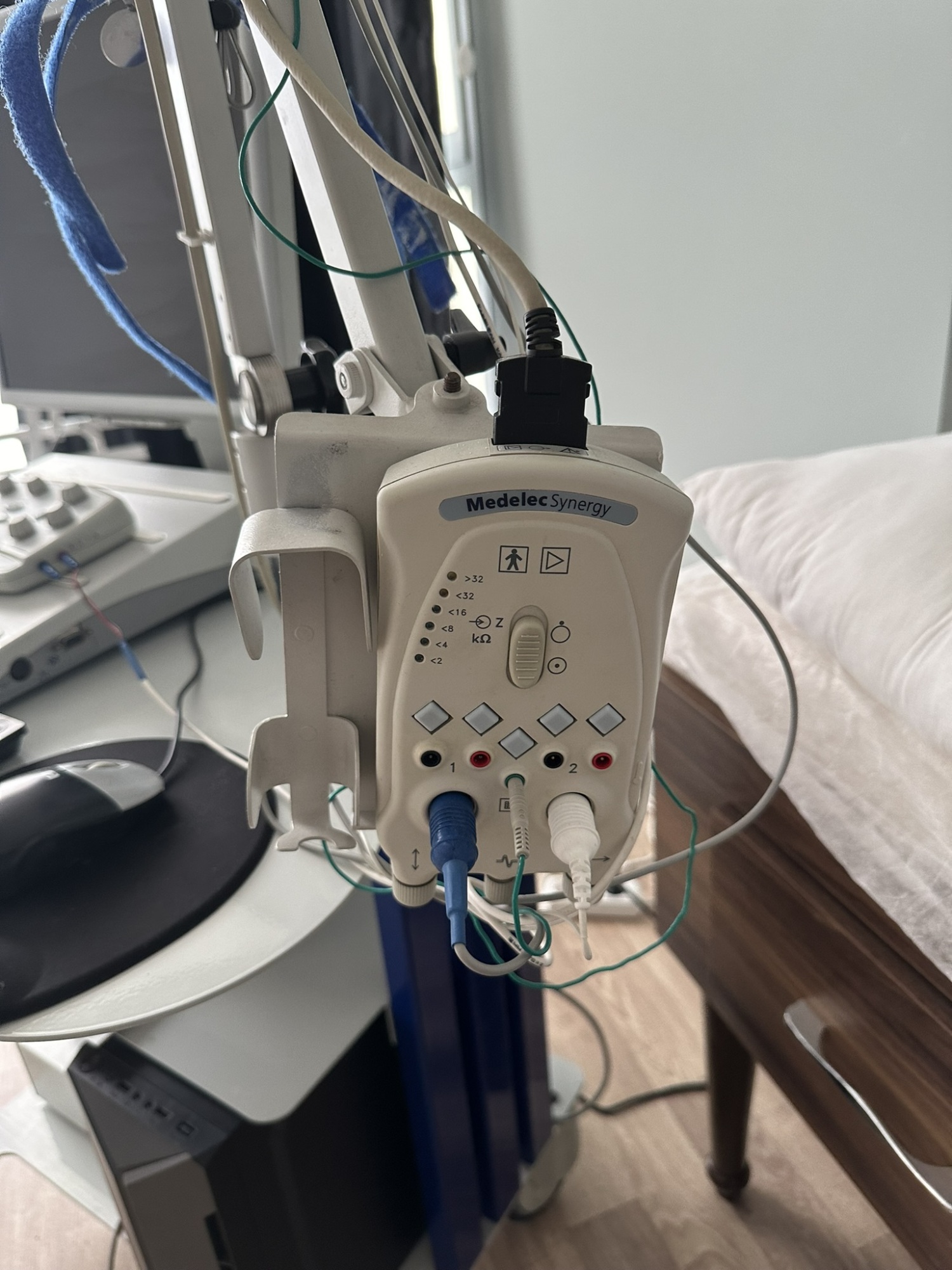
- 1 Medelec Synergy branded amplifier head
- 1 Medelec Synergy branded footswitch
- 1 NEC branded monitor
- 1 black computer case
- 1 black keyboard
- 1 black mouse and mouse pad
- 1 white printer
- 1 orange power supply or isolation transformer
- Various connection cables and electrode wires
Purpose of Use and Authenticity
This device is an electromyography (EMG) system used to assess the health of muscles and the motor neurons that control them. It is used in the fields of neurology and physical medicine for diagnosing nerve entrapments, muscle diseases, and other neuromuscular disorders. The CE and ETL certification labels on the device, the quality of the logo, and the overall workmanship give the impression that the product is an original medical device.
General Condition and Physical Deformation
Although the device is used, it appears to be in good condition overall. No serious physical damage, cracks, or fractures are observed on the parts. There is slight yellowing on the white plastic components, particularly in the area where the labels are located, due to age. There is no significant dirt, rust, or discoloration on the device as a whole. The carrier stand and wheels appear clean and functional.
Mechanical and Electronic Components
The mechanical parts of the system are visually in good condition. The articulated arm of the carrier stand appears sturdy and holds the amplifier head stably. No issues are observed with the mobility of the wheels. All three pedals of the footswitch are in place and show no physical damage. The buttons on the control unit and the indicator lights on the amplifier head are physically intact. As the device’s screen is off, no comment can be made on whether the electronic components are operational.
Accessories and Connections
The system is seen with the main accessories necessary for its operation. The keyboard, mouse, monitor, footswitch, and main control units are connected to the system. The amplifier head has various electrode inputs (where blue, white, and green colored wires are connected) and a main connection socket. All socket inputs and cables are visually in good condition. It cannot be determined if any parts are missing without a standard inventory list for the device, but the basic setup appears to be complete.
Label Information and Technical Details
The label on the underside of one of the device’s components contains the following information:
- Brand: VIASYS Healthcare
- Model Number: 040C004
- Serial Number: 070393
- Year of Manufacture: 2007
- Certifications: CE 0088, ETL Listed (Compliant with UL STD 2601-1 and CAN/CSA STD C22.2 NO. 601.1)
- Warning: “CAUTION – DO NOT REMOVE COVERS. REFER SERVICING TO QUALIFIED PERSONNEL.”
Potential Risk Assessment
Based on visual inspection, there is no obvious evidence (such as broken parts, crushed cables, burn marks, etc.) that would pose an immediate risk of malfunction. The fact that the device was manufactured in 2007 is a factor to consider regarding the lifespan of its electronic components. However, the device’s overall good physical condition for its age suggests that it may have been regularly maintained. Tidying up the cables could reduce stress on the connection points and prevent potential contact issues in the future. In its current state, the device does not exhibit any visual condition that would impede its operation.