Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 02.04.2026
Report code: 1775128607
Fetalcare FC 700 Fetal Monitor Analysis Report
Device Identification and Application Areas
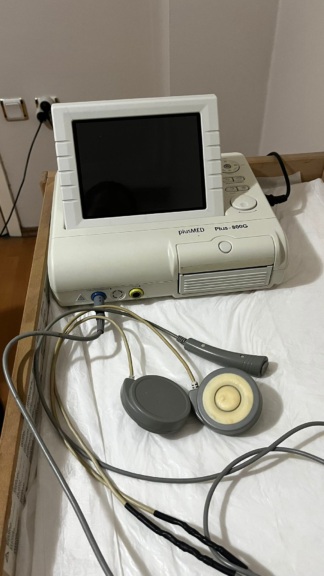
The device in the images is a fetal monitor (NST device) used to monitor the fetal heart rate (FHR) and the uterine contractions (UC) of the expectant mother during pregnancy. It is suitable for use in gynecology and obstetrics outpatient clinics, and in hospital delivery rooms.
Brand and Model Information
Based on the inscriptions located on the device’s front panel and in the quick guide section on the left side, its brand has been identified as Fetalcare, and its model as FC 700.
General Condition and State
The device is in used condition. While slight dust and superficial dirt are observed on the casing, there are no breaks, cracks, or deep scratches that would compromise the device’s integrity. The device’s power (POWER) light is illuminated green, indicating that electrical current is reaching the device.
Mechanical and Physical Examination
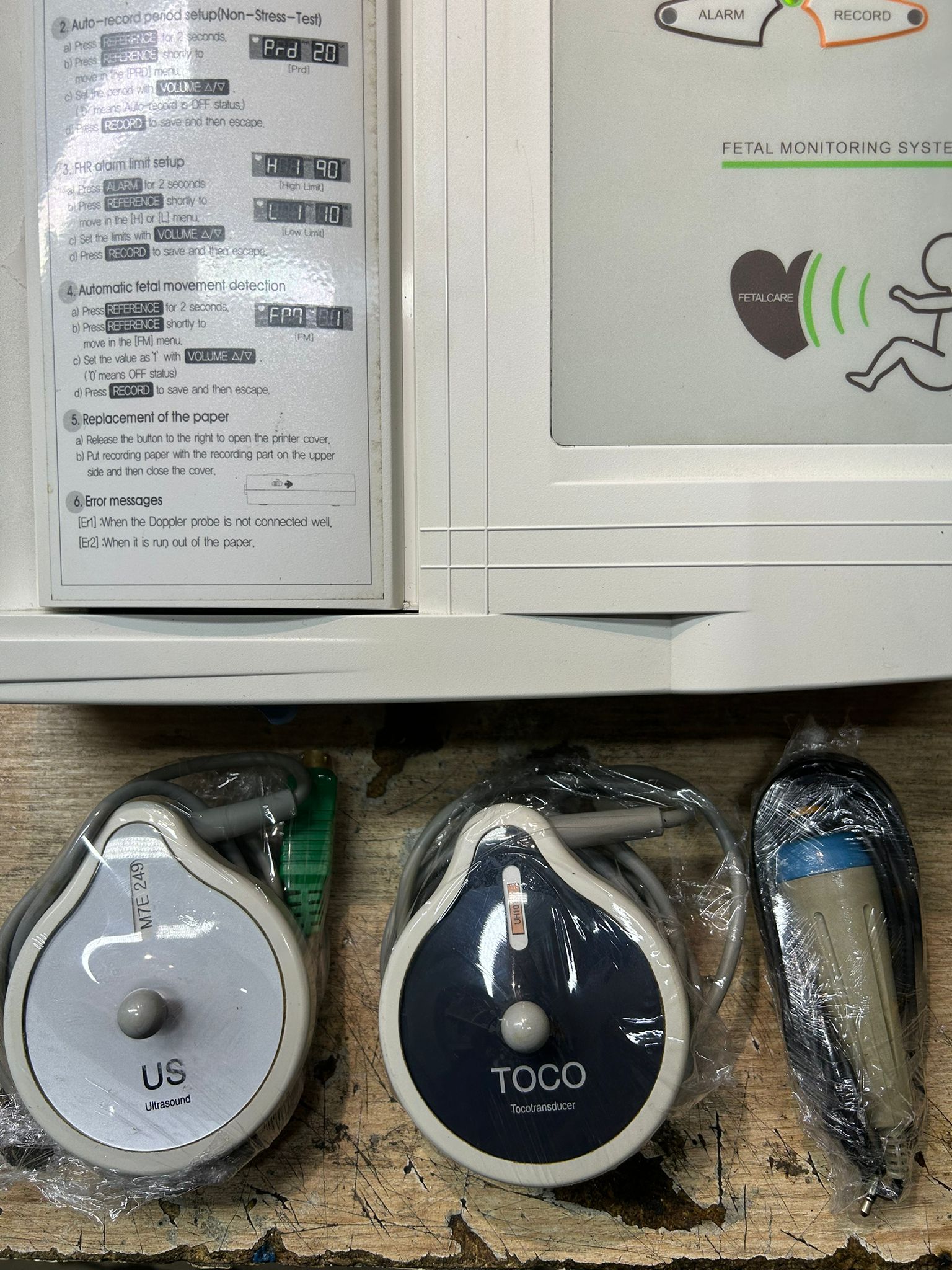
The device’s outer casing is made of white plastic. No wear, punctures, or depressions are observed on the membrane keypad (VOLUME, REFERENCE, ALARM, RECORD) on the front panel. On the left side, there is an integrated thermal printer cover with the device’s quick user guide on it. The guide’s text is clear and legible.
Screen and Electronic Components
The device’s front panel features digital display screens designed to show FHR and UC values. As of the moment the images were taken, despite the power light being on, no numerical data or error codes are displayed on the digital screens.
Accessory Analysis and Quantity Information
In addition to the main device, a total of three accessories wrapped in transparent protective nylon have been identified in the images.
- US (Ultrasound) Probe: It is white and gray, with a circular shape. There is a label on it with M7E 249 written. At the end of its cable, there is a green, multi-pin connection socket.
- TOCO (Tocotransducer) Probe: It is dark navy blue and gray, with a circular shape. There is a partially legible orange label on it.
- Patient Button (Event Marker): It is a gray, cylindrical hand button with a blue stripe. At the end of its cable, there is a standard type metal jack input.
Label and Guide Information
On the device’s left panel, there is an FC 700 Quick Guide (Hızlı Kullanım Kılavuzu) printed directly on the casing. This guide contains the following information.
- Basic usage steps and probe placement diagrams
- Automatic recording duration setting (Non-Stress-Test)
- FHR alarm limit setting
- Automatic fetal movement detection setting
- Paper replacement instructions
- Error messages (Er1: When the Doppler probe is not well connected, Er2: When paper runs out)
Authenticity Assessment
Considering the device’s casing quality, the structure of the membrane keypad, the clarity of the prints on the front panel, and the industrially standard socket structures of the accessories, the product is assessed to be an original medical device.
Potential Malfunction Risk
No physical damage, cable breaks, or crushed sockets that would prevent the device from operating have been detected in the images. The fact that the accessories are in nylon packaging indicates that they are protected from external factors. The device’s general condition is good, and no significant malfunction risk is observed based on the visuals.