Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 30.01.2026
MADSEN AccuScreen Hearing Screening Device Analysis Report
Device Identification
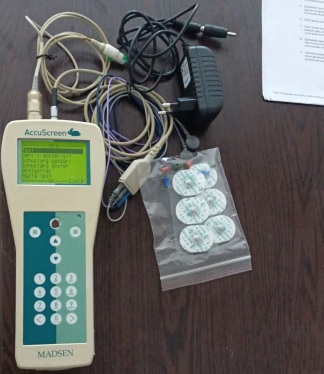
Analysis of the visuals revealed that the product under examination is a handheld medical screening device and its corresponding desktop charging unit (docking station). The device is a portable unit featuring an LCD screen, control buttons, and a numeric keypad. The accompanying part has a slot where the device is placed vertically.
Brand and Model
On the front face of the device, directly above the screen, the clear and legible inscription AccuScreen is present. Additionally, the MADSEN brand name is written in capital letters on the bottom part of the device. In light of this data, the product’s brand has been confirmed as MADSEN and its model as AccuScreen. The product is a professional audiology device used in newborn hearing screening.
Originality and Areas of Use
The brand logos, keypad layout, screen placement, and specific connection ports (ABR, Ear Probe) on the product indicate that it is an original MADSEN medical device. No inscription suggesting questionable originality has been found.
Areas of Use: This device is primarily used in hospital audiology clinics and maternity wards. It is designed to perform hearing screenings (OAE and ABR tests) for newborn babies. The device measures through an ear probe and electrodes.
Connection Ports and Technical Interfaces
The second visual shows the details of the device’s connection panel. The inputs and labels located on this panel are as follows:
- IR Window: Infrared window section used for data transfer or communication.
- ABR Electrodes: 4-pin electrode connection port used for Auditory Brainstem Response (ABR) testing.
- Ear Probe: Multi-pin (approximately 8-10 pins) circular connection port used for connecting the ear probe.
- Bottom Connector: In the first visual, a metal-pinned socket input is visible at the very bottom of the device, used for the charging unit or PC connection.
Overall Condition and Physical State
The cosmetic condition of the device has been assessed as “used.” There are noticeable signs of wear on the outer casing due to use. Yellowing (discoloration) of the plastic casing over time is observed; the device’s original beige/gray color has shifted to a slightly yellowish hue. Dust/dirt accumulation is present at panel joints and corners.
Physical Deformation and Mechanical Components
No deep cracks or fractures have been detected on the device’s outer casing. However:
- Slight abrasions are present on the upper left and upper right frame sections of the screen.
- Color tone differences and minor stains are visible on the plastic surface of the charging unit.
- Although the keypad panel appears intact, the legibility of the inscriptions on the keys has been preserved. There is no excessive wear on the keys.
- Dusting and slight darkening are present on the edges of the panel containing the port inputs (second visual).
Screen Analysis
As the device is in the off position, the condition of the screen’s pixels, lighting capacity, or any error messages cannot be displayed. Physically, no deep scratches or cracks are visible on the screen glass.
Accessories and Quantity Information
A total of 2 pieces of product are present in the visuals:
- 1 unit of MADSEN AccuScreen handheld device.
- 1 desktop charging/docking unit.
Missing Parts: The ear probe, electrode cables, power adapter (charger cable), and consumables required for the device’s operation are not present in the visuals. Only the main unit and the dock stand are available.
Battery Status
As the device is shown with a charging station (dock), it is understood to have a rechargeable internal battery. However, since the battery is located inside the device, its physical condition (swelling, leakage, etc.) cannot be observed externally. Considering the device’s manufacturing technology and cosmetic condition, there is a high probability that the battery may require replacement.
Potential Malfunction Risk
Visual inspection did not reveal any major damage compromising the device’s external integrity. However, the exposed nature of the device’s connection ports (especially Ear Probe and ABR sockets) and the accumulation of dust on the edges may suggest a risk of oxidation at the contact points. Considering the device’s age and the condition of the plastic material used, there is a possibility that its electronic lifespan may have ended or that it may require calibration.