Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 13.10.2025
B. Braun Diapact CRRT Hemodialysis Device Analysis Report
Overview and Device Identification
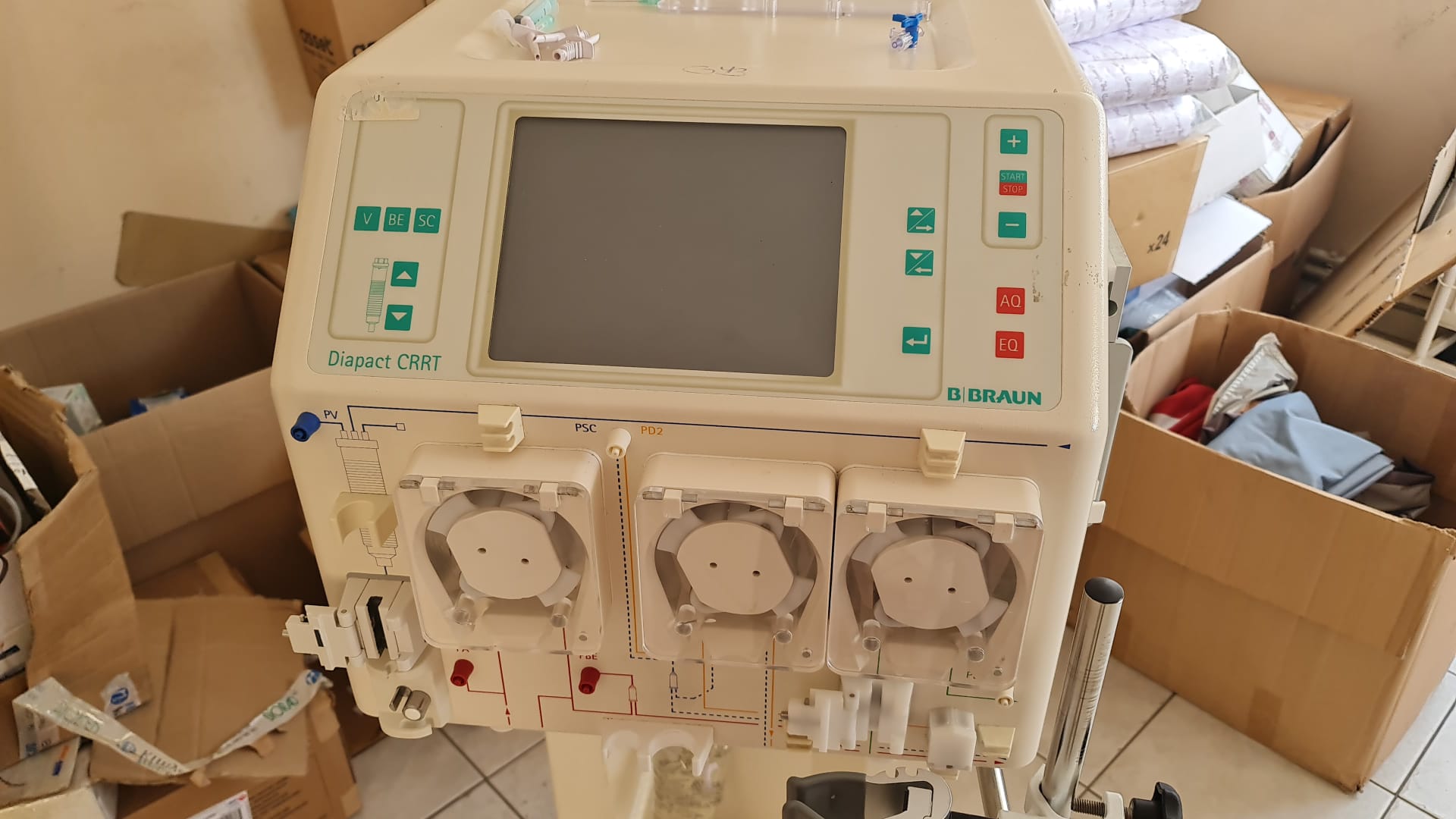
The product shown in the visuals is a medical device designed for Continuous Renal Replacement Therapy (CRRT). The device’s front panel features a screen, control buttons, status indicators, and three peristaltic pumps. The purpose of the product is to clean the blood outside the body in patients with kidney failure.
Brand and Model Information
During the inspection, the brand and model information of the device was clearly confirmed from both the writing on its front face and its rear label.
- Brand: B. Braun
- Model: Diapact CRRT
- Manufacturer: B. Braun Avitum AG, Melsungen, Germany
Physical Condition and Deformation
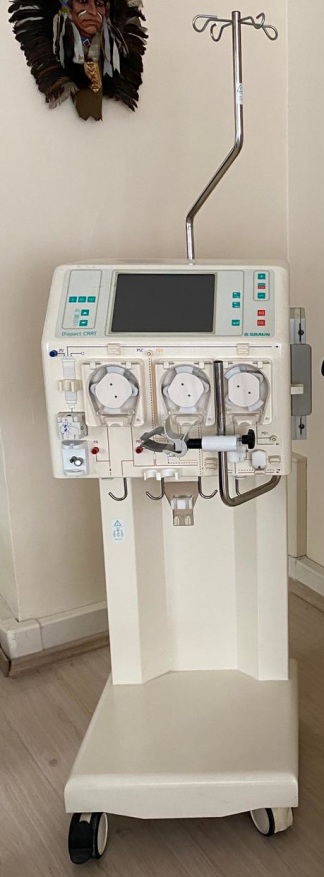
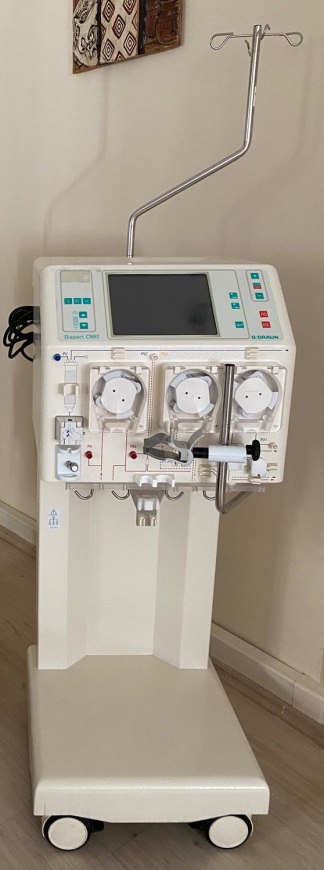
The device is generally a used product. No significant dents, breaks, cracks, or crushes were detected on its outer casing. However, a general discoloration (yellowing) is observed in the plastic components, which may indicate its previous use or storage conditions. The device being surrounded by boxes suggests it is in a storage environment. There is no significant wear on the front panel and buttons.
Mechanical and Electronic Components
Upon visual inspection of the mechanical parts, the three pump mechanisms and their covers on the front panel appear complete. No breaks or deformations were detected in the beds where the tubes are attached. Physical buttons such as “START/STOP”, “+”, “-“, “AQ”, and “EQ” are present on the control panel. The device’s screen is off, therefore it cannot be determined if there are any error messages or images on the screen, and no comment can be made regarding the functionality of the electronic components.
Label and Technical Information
The manufacturer’s label on the rear of the device contains important technical details and identification information related to the product. This information is as follows:
- REF Code: 7106505
- Serial Number (SN): O3520
- Manufacturing Date: 2016-04-18
- Voltage Range: 110/240V
- Frequency: 50/60 Hz
- Power Consumption (VA): 800
- IP Protection Class: IP 21
- CE Approval Code: CE 0123
- GTIN Code: 04039239041238
Additionally, the label includes an “Rx only” warning stating that the product should not be used in environments with flammable anesthetic gases and can only be used with a prescription.
Accessories and Quantity Information
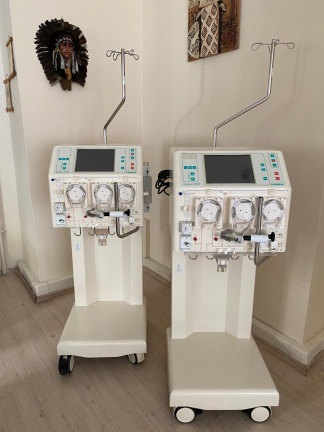
The visuals show one B. Braun Diapact CRRT device. The power cable, complete tubing sets, or other external accessories necessary for the device’s operation are not visible in the photographs. Only short tubing pieces attached to the pump mechanisms are present. The front section of the device features connection ports for various fluid lines and sensors.
Potential Risk Assessment
Based on visual inspections, there is no significant damage or wear on the device’s casing or mechanical components. This indicates that the device is physically well-preserved. However, due to the device being unpowered and its screen off, it is unknown whether the electronic components are functional. While it does not pose an obvious risk of malfunction in its current state, as with any used medical device, it is recommended that it be checked and calibrated by an authorized technician before use.