Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 22.04.2026
Report code: 1776861401
Medtronic MiniMed Insulin Pump and Accessories Analysis Report
Device Identification
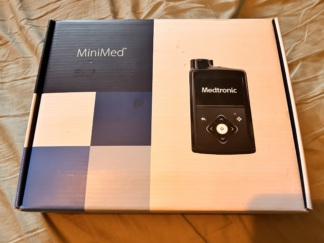
The images show one insulin pump device, its original box, and related consumable accessories.
Brand and Model
The information on the device and its box is clearly legible. The brand of the product is Medtronic, and the model series is MiniMed. The “MiniMed™” logo is clearly visible on the box, and the “Medtronic” logo is clearly visible on the device.
Authenticity
The box design of the product in the images, the quality of the logos on the device, the layout of the labels, and the general workmanship details indicate that the product is an original Medtronic product.
Usage Areas
This device is an external insulin pump used in the insulin therapy of diabetic patients. It provides continuous and adjustable doses of insulin delivery to the body. It is suitable for use in the field of endocrinology for the treatment of type 1 diabetes and some type 2 diabetes patients. The accessories in the images (reservoir and infusion set) are complementary parts necessary for the operation of this pump and the delivery of insulin to the patient.
Quantity Information
A total of the following products have been identified in the images:
- 1 Medtronic MiniMed insulin pump device.
- 1 original outer box of the device.
- 1 inner cardboard compartment for the device.
- 1 MiniMed Quick-set infusion set box.
- 1 MiniMed Reservoir box.
- 1 black belt clip or protective case part.
- 1 blue apparatus in a transparent bag (likely a tubing clip).
- 1 multi-language warning/information leaflet.
General Condition
The general condition of the device and its accessories appears new and unused. The products are in their original packaging, and their placement within the box remains undisturbed. The screen and body of the device are clean.
Physical Deformation
No scratches, dents, cracks, discoloration, or dirt have been detected on the device’s casing, screen, or buttons. No significant deformation caused by shipping or storage is observed on the outer box or the accessory boxes.
Mechanical Components
The mechanical parts of the device (reservoir compartment cover, battery compartment cover, buttons) appear sturdy and in place. No broken or missing parts are noticeable.
Electronic Components
The electronic components of the device appear intact as far as can be seen from the outside. The screen surface is smooth. There is no visual evidence regarding whether the device is in working order (whether it turns on or not).
Accessories
The accessories included with the device are as follows:
- MiniMed Quick-set: Infusion set box.
- MiniMed Reservoir: Insulin reservoir box.
- A black clip/case part.
- A blue apparatus in a transparent bag.
The expiration dates on the accessory boxes are not clearly legible, but the boxes appear closed and new. There is a reservoir connection socket and a battery compartment cover on the top of the device.
Battery Status
It cannot be determined from the images whether the device has an internal battery. However, a battery compartment cover is visible on the top of the device, indicating that it operates with an external battery (e.g., AA or AAA). No signs of leakage or corrosion are visible in the battery compartment.
Label Information
Specific information such as serial number, lot number, or REF code on the labels of the device or boxes is not clearly legible. Only model names and manufacturer information are present on the accessory boxes.
Screen Analysis
The device screen is in the off state. Therefore, no text, warnings, or menus are visible on the screen.
Size and Compatibility
The size of the device is consistent with a typical insulin pump and is portable. The accessories (Quick-set and Reservoir) are compatible with Medtronic MiniMed series pumps.
Year of Manufacture
No clear information regarding the year of manufacture of the product could be determined from the labels or boxes in the images.
Documents
No invoice or warranty certificate is visible in the images. Only a multi-language warning/information leaflet is present.
Usage Duration
There is no information regarding the usage duration (operating hours, etc.) of the device in the images. The new appearance of the device suggests that it has not been used.
Existing Faults
There is no clear evidence in the images, such as broken parts or error messages, indicating that the device is faulty.
Potential Risk of Failure
No significant wear, rust, or damage has been observed on the device in the images. The condition of the product appears to be very good. Therefore, based on the visual data, no potential risk of failure is foreseen.