Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 25.03.2026
Report code: 1774421047
Medtronic MiniMed Insulin Pump Kit Analysis Report
Device Identification and Usage Areas
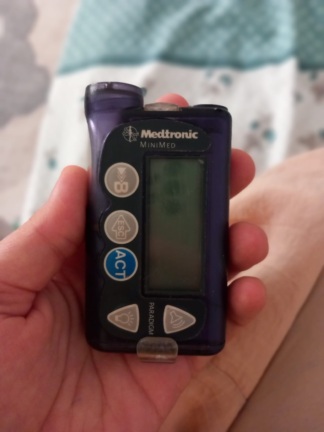
The product shown in the images is a comprehensive kit containing an insulin pump used in diabetes management, along with various consumables and accessories for this pump. The device is designed to provide continuous and programmable insulin infusion into the patient’s subcutaneous tissue.
Brand and Model Information
Upon examination of the main device box and documents, it has been determined that the brand of the product is Medtronic and the series is MiniMed. One of the accessory labels features the designation 640G.
General Condition and Authenticity
The product set is generally in brand-new and unused condition. The main insulin pump box and the majority of the accessories are contained within their original factory seals and sealed blister packaging. The packaging quality, box designs, logo prints, and barcode label standards clearly indicate that the product is authentic.
Box Contents and Quantity Information
The products and accessories identified in the images are listed below.
- One sealed, shrink-wrapped Medtronic MiniMed insulin pump box
- One four-pack of Energizer MAX AA batteries
- One two-pack of Energizer Industrial AA batteries
- One large white consumables box labeled Medtronic MiniMed
- One transparent infusion set component in sealed blister packaging
- One triple reservoir set with blue plungers in sealed blister packaging
- One set of blue and white tubing clamps in a sealed bag
- One black pump clip in a sealed bag
- One transparent protective component in a sealed bag
- One black spare part in a sealed bag
- One English warning and information document
Physical and Mechanical Evaluation
Since the main device is inside a sealed box, it is not open to direct physical contact or observation. According to the image on the box, the front surface of the device features a screen and navigation buttons. There is no crushing, tearing, discoloration, dirt, or physical deformation on the main box housing the device, the accessory packaging, or the consumables. All packaging maintains its structural integrity.
Battery Status
The set includes the external batteries required for the operation of the device. The battery packaging is sealed. No signs of leakage, swelling, or oxidation are visible on the batteries.
Label and Reference Information
The label information found on the accessory packages is stated below.
- Pump Clip: REF ACC-1601, LOT 111605279, manufactured in Costa Rica.
- Activity Guard: REF ACC-1520, LOT A2025084, manufactured in Singapore.
- Tubing Clamps Kit: Coded 7005153-004_C, with the designation 640G 21L present on the label.
Documents and Warnings
The set includes an illustrated caution document explaining the compatibility of insulin pump series with battery caps. In the document, the correct usage combinations of old and new model pumps with different battery caps are visualized with green checkmarks and red cross signs.
Potential Failure Risk
As all products are in their sealed packaging and in brand-new condition, there is no potential failure risk or wear and tear identifiable through the images. The product set is complete, undamaged, and in excellent condition.