Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 10.02.2026
Medtronic MiniMed Insulin Pump and Accessories Analysis Report
Device Identification and Purpose of Use
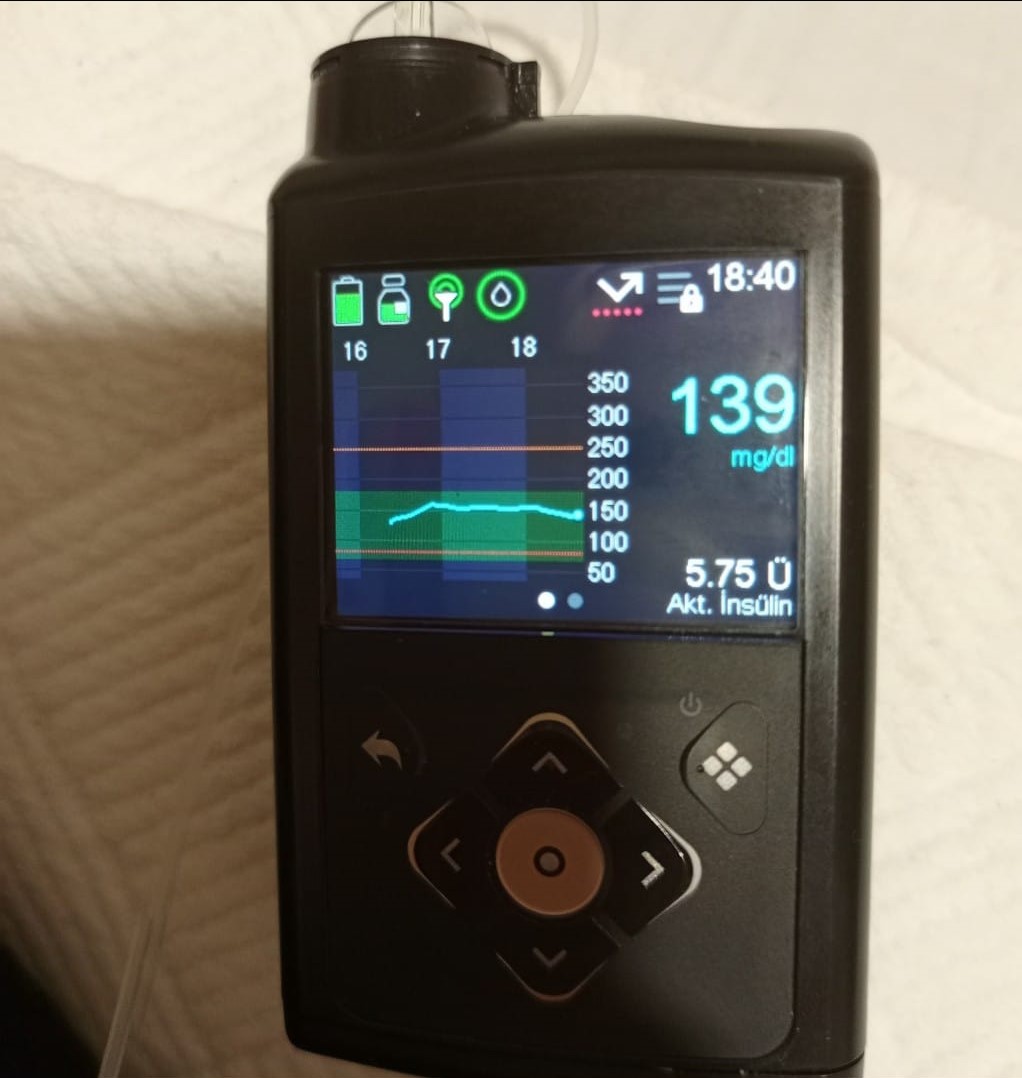
The product examined in the visuals is an insulin pump with advanced electronic hardware, used in diabetes (sugar disease) management. This device is a vital medical equipment designed to deliver insulin continuously or in programmed doses to the user’s body. The product is used to ensure blood sugar regulation for diabetic patients, based on sensor data monitored via its digital screen.
Brand and Model Information
The information obtained as a result of detailed examination of the product in the visuals and verification of the codes on its rear label is as follows:
- Brand: Medtronic
- Product Family: MiniMed™
- Model Identification: Based on technical verification, taking the device’s interface design (Blue shield icon, graphical structure), Bluetooth feature, and the codes “REF M994838A002” and “Anatel 03467-22-02516” on the rear label as reference, the product has been identified as an insulin pump belonging to the Medtronic MiniMed 780G system.
- Interface Language: The phrases “Akt. İnsülin” (Active Insulin), “İnsülin” (Insulin) and the unit display “mg/dl” in the screen visuals confirm that the device’s software language is Turkish.
Cosmetic and Physical Condition
Detailed observations regarding the product’s casing and general condition are as follows:
- Screen Condition: There is a transparent factory protective film on the device screen. Air bubbles and liftings are observed on the edges of the film (especially in the bottom corners). However, this condition belongs to the film, indicating that the screen itself is clean and undamaged underneath, likely suggesting a near-new or very cleanly used device. No pixel errors or cracks are visible on the screen.
- Casing Structure: No deep scratches, dents, or impact marks were found on the black plastic outer casing. The keypad (directional keys, selection key, and menu keys) appears clean, with no fading or deformation.
- Waterproofing: The device’s rear label includes the inscription IPX8, which indicates that the device is water-resistant and its physical integrity (seals, etc.) should be preserved. No openings were visually detected in the casing.
Screen Analysis and Operational Data
The device is displayed in operational status. The data on the screen summarizes the current situation as follows:
- Glucose Value: 139 mg/dl (Sensor data).
- Active Insulin: Displayed as 5.75 U (Units).
- Connection and Warnings: The top information bar includes a “Lock” icon (keypad lock active), battery level icon (green, full), reservoir status icon (nearly full), and a signal interruption indicator (connection/sensor signal status).
- SmartGuard Feature: The blue shield icon and graphical interface indicate that the device uses SmartGuard technology and has automatic mode features.
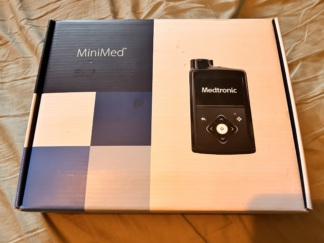
Accessories and Package Contents
In addition to the device, the box contents include the following original accessories:
- 1 Activity Guard: In its original sealed packaging (with Medtronic logo). Label Information: REF ACC-1520. This part is used to prevent accidental presses of the device’s buttons or to secure it with a clip.
- 2 Blue Clips: Spare accessories in a sealed bag, with the Medtronic logo, used to secure tubing or connections.
- 1 Belt Clip (Holster): A black hard plastic attachment that allows the device to be worn on a belt (with DT and recycling PS logos). Placed in the box slot.
- Infusion Set (Partial): A thin transparent tube (infusion set line) attached to the device is visible in one of the visuals, but the complete set or needle tip is not visible.
Label and Technical Data (Device Rear and Packages)
The critical serial and production codes identified during the analysis are as follows:
- REF Code (Device): M994838A002 (Technical model reference of the device).
- Anatel Code: 03467-22-02516 (Indicates 2022 homologation, a modern device).
- Lot Number (Accessory): The inscription “LOT A2024228” was read on the ACC-1520 packaging.
- Certifications: CE 0459 (European Conformity), IPX8 (Water resistance), WEEE (Recycling) marks are present.
- Contact: “Medtronic MiniMed Northridge, CA 91325 USA” address is stated as the manufacturer.
Quantity Information
Total number of parts identified within the package:
- 1 Medtronic Insulin Pump (Main Device)
- 1 Belt Clip (In the slot under the device)
- 1 Pack of Activity Guard (Sealed packaging)
- 1 Pack of Spare Blue Clips (Sealed packaging, with 2 pieces inside)
- 1 Device Box (Black, foam-protected)
Battery and Power Status
The device operates with AA alkaline batteries. The battery icon in the upper left corner of the screen is at full (green) level. No low voltage warning for the battery is visible on the screen. The battery compartment cover (the protrusion seen in the upper left of Image 2) is robust and closed, with no signs of leakage or corrosion visible externally.
Conclusion and Authenticity Assessment
The examined product is a 100% original Medtronic brand medical device. The presence of the protective film on the screen, the accessories (Activity Guard, etc.) being in their unopened sealed bags, and the cosmetic cleanliness of the device strongly indicate that the product is either in “near-new, very lightly used” or “open box” condition. The Turkish interface is a significant advantage for users in the Turkish market. There are no visible breaks, cracks, or “Service” error warnings on the device that would hinder its operation.