Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 31.07.2025
Medtronic MiniMed 670G Insulin Pump and Accessory Analysis Report
Product Identification and General Overview
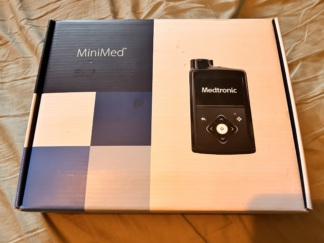
The images show one insulin pump and one blood glucose meter used for diabetes management. The products are identified as the Medtronic MiniMed 670G insulin pump, as indicated on the box, and a blood glucose meter compatible with this system. The insulin pump is presented with its original box.
Brand and Model Information
Upon inspection, the brand and model information of the devices has been identified as follows:
- Insulin Pump Brand-Model: Medtronic MiniMed 670G
- Blood Glucose Meter Brand-Model: The device in the image resembles a Contour Next Link 2.4 model, which is used for wireless data transmission with Medtronic pump systems. This information is an estimation based on the device’s design and its known compatibility with the MiniMed 670G.
Area of Use and Functionality
The Medtronic MiniMed 670G is an insulin pump designed to regulate blood sugar by continuously delivering small doses of insulin to the body. The accompanying blood glucose meter measures glucose levels from a blood sample taken from the finger and wirelessly sends this data to the insulin pump for calibration or bolus calculations.
Quantity and Content Information
The products shown in the images are as follows:
- 1 Medtronic MiniMed 670G Insulin Pump
- 1 Blood Glucose Meter
- 1 Medtronic MiniMed 670G Original Box
Additional accessories such as a charger, user manual, or other consumables are not visible in the images of the box contents.
General Status and Condition Assessment
Both devices are generally in good condition and appear to be lightly used. The cleanliness of the products and the pump’s presence in its original box suggest it was well-maintained. No serious physical defects have been observed.
Physical and Mechanical Condition
There are no significant deformations such as scratches, dents, cracks, discoloration, or dirt on the casings, screens, or keypads of the devices. The mechanical components of the pump, such as the reservoir cap and buttons, appear visually intact and problem-free.
Electronic Components and Screen Analysis
As the insulin pump’s screen is off, an assessment of its operational status cannot be made. However, the blood glucose meter’s screen is on, and its menu is clearly visible. Options such as “MENU”, “Logboek” (Logbook), “Trends”, and “Bolus” are clearly legible on the screen. This indicates that the device’s electronic components are active.
Label and Manufacturing Information
Upon examining the label on the product’s original box, the following information was obtained:
- REF Code: MMT-1782K
- Serial Number (SN): NG1853358H
- Date of Manufacture: 2019-03-06
- Expiration Date: 2022-03-05 (It is likely that this date applies to a sterile accessory inside the box rather than the pump itself.)
- Lot Number/Other Code: HG383LX
The label information contains important details that confirm the product’s authenticity.
Potential Malfunction Risk
In the images, no findings that could negatively affect the operation of the devices, such as any rust, damage to cables, severe wear, or broken parts, were observed. Based on the current physical condition of the devices, a risk of malfunction in the near future is not anticipated.