Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 21.04.2026
Report code: 1776770731
Medtronic MiniMed Paradigm Insulin Pump Analysis Report
Device Identification and Credentials
The device in the images is a medical insulin pump used in diabetes treatment. The device features a transparent plastic casing, control buttons, and a display screen. The brand and model information can be clearly read from the label on the back surface.
- Brand: Medtronic MiniMed
- Model: Paradigm
- REF Code: MMT-715WWL
- Serial Number: SN PBR366068H
- CONF Code: CONF EB96
Usage Areas and Purpose
This device is used to provide continuous and programmable insulin delivery throughout the day for patients with type 1 diabetes or patients with type 2 diabetes who require insulin. It is suitable for patients under the follow-up of the endocrinology department. The device delivers insulin through an infusion set (not present in the images) attached to the patient’s body.
General Condition and Physical Deformation
The general condition of the device appears to be good. No deep scratches, cracks, or fractures have been detected on the transparent casing. The screen glass is intact and readable. The text and symbols on the control buttons are clearly visible. There is no significant dirt or dust accumulation on the outer surface of the device. The label on the back surface is intact, and the information on it is legible.
Mechanical and Electronic Components
The structural integrity of the casing, which constitutes the mechanical assembly of the device, is preserved. The battery cover (top right) is in place. The reservoir compartment (bottom left) where the infusion set is attached appears empty. The display screen, which is part of the electronic assembly, is functional and capable of displaying different menus. The control buttons appear physically intact. Some wires (yellow and red) inside the device are visible through the transparent casing and appear to be intact.
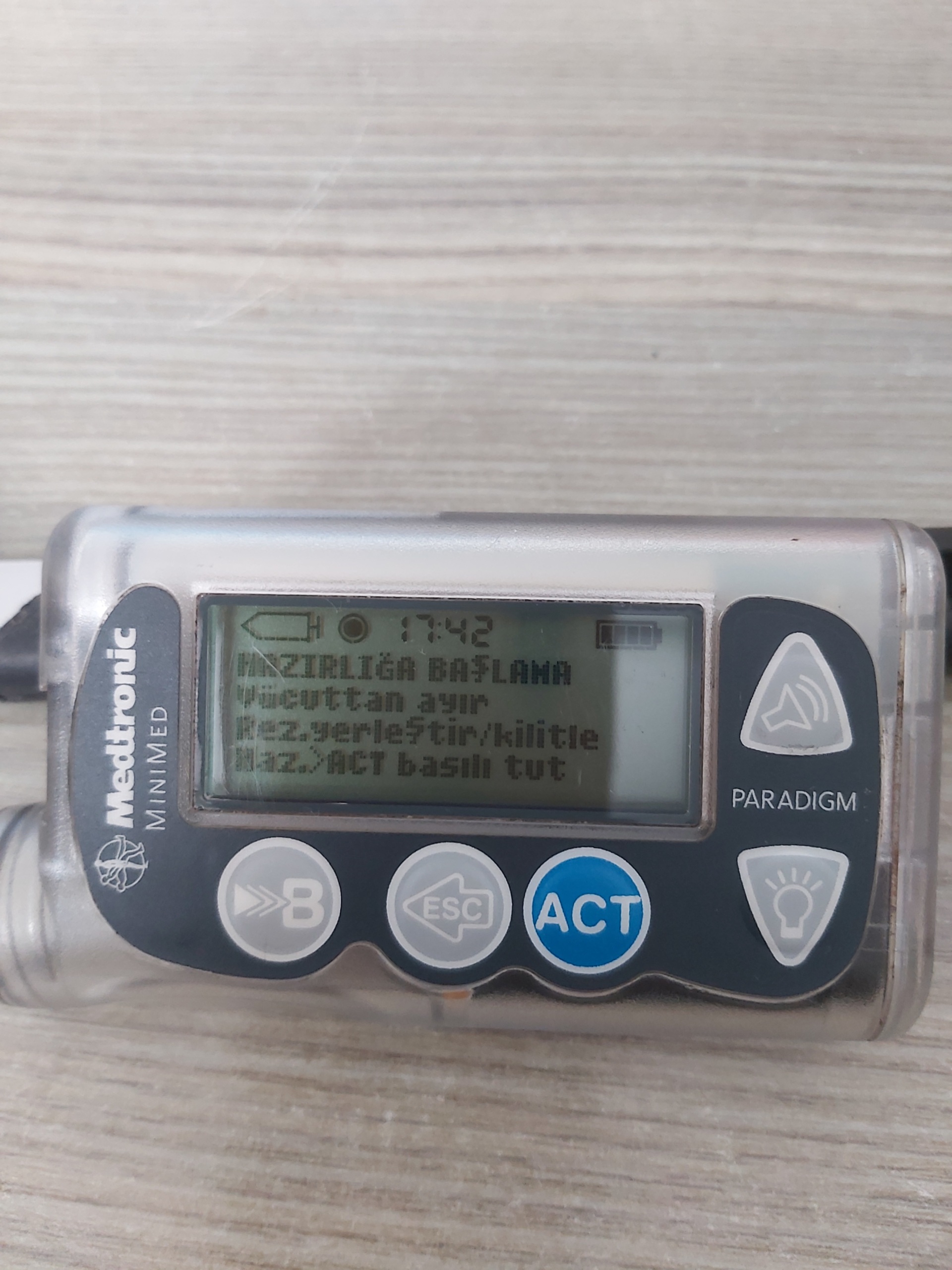
Screen Analysis and Menus
The device screen operates with the Turkish language option. The following screen captures have been identified in the various images:
- Image 1: Warnings such as “START PREPARATION”, “Disconnect from body”, “Insert/lock res.”, “Prep > Hold ACT” are visible. This indicates that the device is in the process of a new reservoir filling and preparation phase. The time is 17:42.
- Image 2: Information such as “STATUS U100”, “Rewind pump”, “Basal 4: 1.70 U/h”, “Empty reservoir” is present. This screen shows the current status of the device, the basal insulin rate, and that the reservoir needs to be emptied. The time is 17:38.
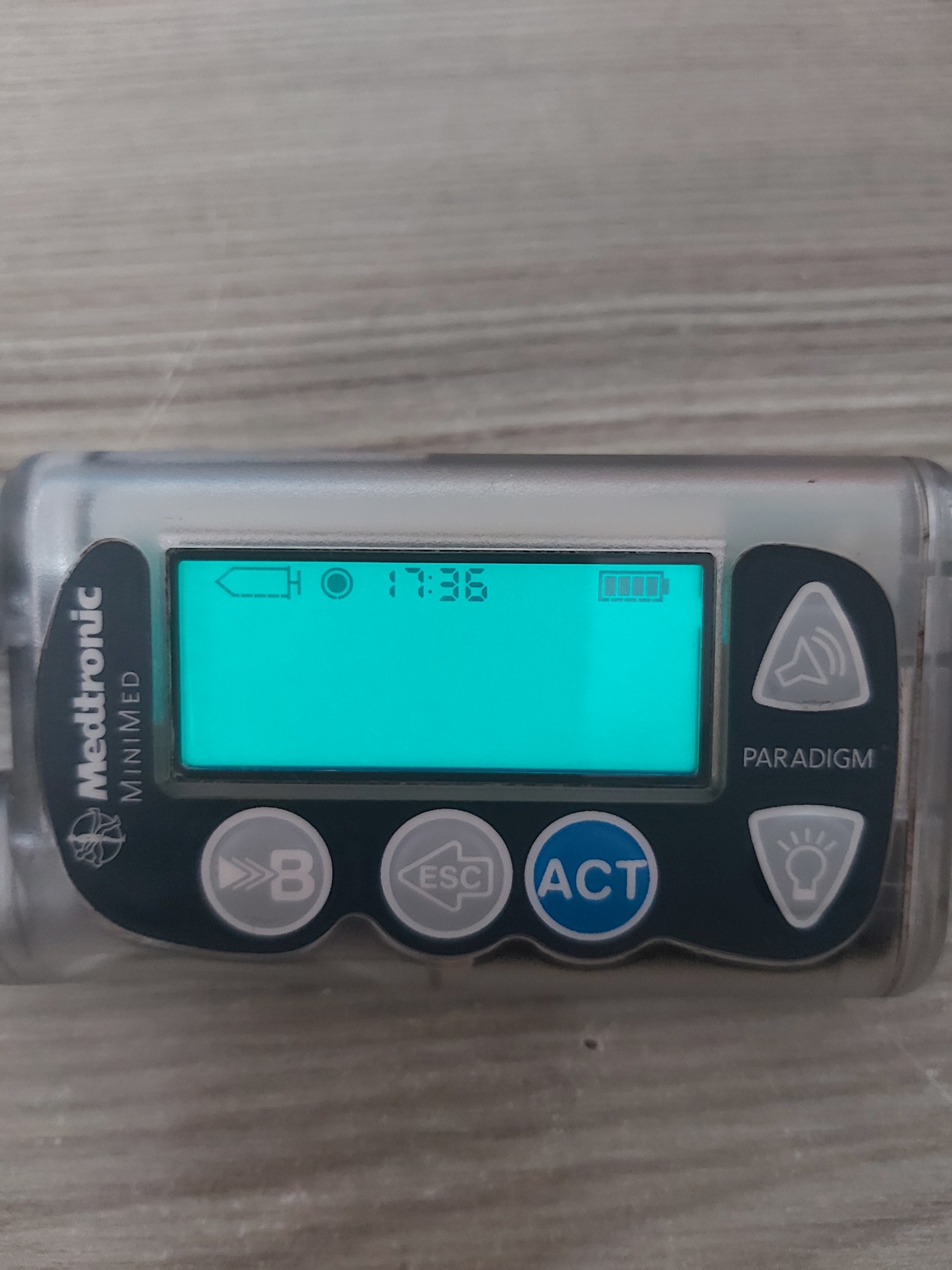
- Image 3: The screen backlight is on (green), but no text is visible. The time is 17:36.
- Image 4: The “SET TIME” menu and the time “17:36” in large font are visible. This indicates that the device is in the time setting menu.
A fully charged battery icon is present in the top right corner of all screens.
Battery and Accessories
The device operates with one AAA (LR03) alkaline battery, as indicated on the back label. The battery compartment is located on the top right of the device. The battery icon on the screen indicates that the battery is full. Apart from the device itself, no accessories (infusion set, reservoir, battery, carrying clip, etc.), box, invoice, or warranty certificate are present in the images.
Technical Information and Label Details
The label on the back of the device contains the following technical information and symbols:
- Manufacturer: Medtronic MiniMed, Northridge, CA 91325 USA
- European Representative: Medtronic B.V., 6422 PJ Heerlen, NL
- Water Resistance: IPX7 (Protected against immersion in water for a specific duration and depth)
- Battery Type: AAA (LR03)
- Symbols: CE mark (notified body number 0459/0976), Type BF applied part symbol, caution symbol, MR unsafe symbol, WEEE symbol (electronic waste), Rx Only symbol.
The manufacturing year or the service life of the device could not be determined from the images.
Fault and Risk Assessment
There is no clear physical evidence (broken parts, leaked battery, etc.) in the images suggesting that the device is faulty. The fact that the screen works and navigation between menus is possible indicates that the electronic components are functional. Since the physical condition of the device is good, the risk of a mechanical failure in the near future appears low. However, as it is a medical device, it is recommended that a full functional test be performed by an authorized technical service before use.