Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 15.04.2026
Report code: 1776259303
Löwenstein Medical Luisa Ventilator Device Analysis Report
Device Identification and Brand Model Information
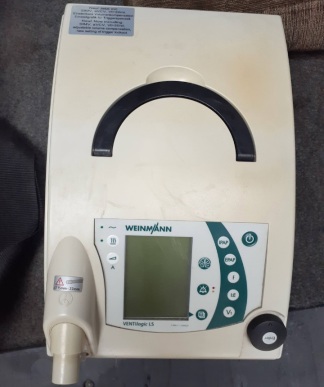
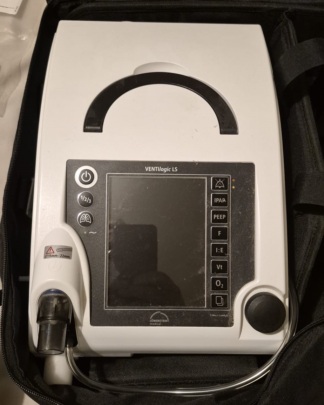
The product in the visuals is a Luisa model ventilator (respiratory) device from the Löwenstein Medical brand. The device’s brand and model are clearly readable on both the label on the front panel and the technical information label on the back. Additionally, a carrying case is included with the device in the visuals.
General Condition and Physical Deformation
The general condition of the device appears to be good. No significant physical damage such as breaks, cracks, or deep dents were detected on its casing or screen. However, there are minor scratches, fingerprints, and dust on the large touchscreen surface and the white plastic casing, consistent with use. The carrying case is clean and appears sturdy.
Mechanical and Electronic Components
The device’s mechanical parts, buttons, and connection ports are intact. The power and alarm mute buttons on the front panel are in place. The respiratory circuit connection ports on the side, sensor inputs (P, SpO2, CO2, Neb), and air outlets appear to be in good condition. The battery compartment cover and filter housing on the back are closed. While the device’s electronic components appear visually sound, a functional test could not be performed as the device is not operational.
Accessories and Socket Inputs
Only one gray carrying case with “BREATHEANYWHERE WITH LUISA” and “LÖWENSTEIN medical” written on it is visible with the device in the visuals. A power cable, respiratory circuits, sensors, or other consumables are not present in the visuals. The device has dedicated socket inputs on its side for various medical sensors and connections.
Label Information
The label on the back of the device contains the following information:
- Serial Number: 50047924
- REF Code: 31410
- Model: LUISA LM150TD
- Manufacturer: Löwenstein Medical Technology GmbH + Co. KG
- Year of Manufacture: 2024-10-29
- Other Information: CE0197, MD IP22, Made in Germany, HFT-Mode, 12 V DC max. 7 A, 24 V DC max. 5.4 A, 48 V DC max. 2.7 A.
Additionally, there is another blue label on the back of the device with “SAPIO” written on it.
Usage Areas and Compatibility
This device is used in the respiratory treatment of patients requiring mechanical ventilation support at home or in a hospital setting. The “HFT-Mode” on the label indicates that it has a High Flow Therapy feature. Although there is no specific text on the device regarding infant, child, or adult compatibility, such ventilators generally cater to a wide patient population.
Potential Risk of Malfunction
There is no clear evidence in the visuals, such as broken parts or error messages, indicating that the device is malfunctioning. The physical condition of the device is good, and no signs of significant wear have been observed. Therefore, based on the visual data, the product’s condition can be described as good.