Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 21.04.2026
Report code: 1776759083
Löwenstein Medical Luisa Ventilator Device Analysis Report
Device Identification
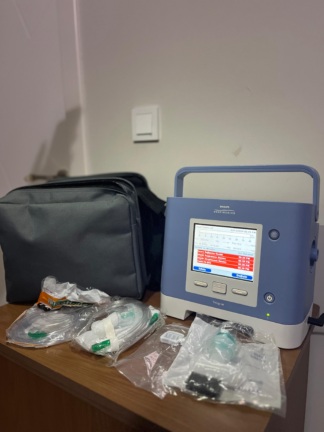
The images show two mechanical ventilator devices and various accessories belonging to them.
Brand and Model
The Löwenstein Medical brand and Luisa model name are clearly legible on the labels on the devices and their carrying cases.
Authenticity
The quality of the logos on the devices, the details of the labels, the serial numbers, and the overall workmanship indicate that the products are authentic.
Usage Areas
The Löwenstein Medical Luisa is a mechanical ventilator device that provides respiratory support to patients. This device is designed for use in hospitals or home care settings for the treatment of patients suffering from respiratory failure.
Quantity Information
There are a total of two ventilator devices in the images. Additionally, there are two carrying cases, one power adapter, one hand pump with a manometer, one external battery, and various packaged filters and accessories.
General Condition
The general condition of the devices and accessories appears to be very good. The products are clean, and the presence of packaged accessories suggests that the devices have been used very little or are in like-new condition.
Physical Deformation
No visible scratches, dents, cracks, or discoloration have been detected on the casings, screens, or other parts of the devices. The plastic components maintain their integrity.
Mechanical Components
The buttons, carrying handles, and socket inputs on the devices appear to be intact. The zippers and stitching of the carrying cases are sturdy.
Electronic Components
Since the screens of the devices are off, there is no visual data regarding their electronic functionality. However, electronic components such as the power adapter and the external battery appear physically intact.
Accessories
The following accessories have been identified in the images:
- Two black carrying cases.
- One power adapter and cable.
- One hand pump with a manometer.
- One external battery.
- Packaged filters and various small parts.
- User manuals.
There are various socket inputs on the devices, and these inputs appear clean. The expiration dates of the accessories cannot be read from the images.
Battery Status
There is one external battery in the images. No signs of swelling or leakage are visible on the outer casing of the battery.
Label Information
The label on the bottom of the device contains the following information:
- REF Code: 31410
- Serial Number: Not clearly legible from the image.
- Year of Manufacture: The symbols on the label indicate the year 2021.
- Other Information: CE 0197 mark, IP22 protection class, and various technical symbols are present.
Screen Analysis
The screens of the devices are off; therefore, no information is available regarding the text or menus on the screens.
Size and Compatibility
The size of the devices indicates that they are of a portable type. There is no clear information on the labels regarding patient compatibility (infant/pediatric/adult).
Year of Manufacture
The year 2021 is indicated next to the factory symbol on the device label.
Documents
User manuals are visible in the images, but no invoice or warranty certificate has been detected.
Usage Duration
There is no information on the screen or label regarding the usage duration of the devices.
Existing Faults
There is no clear evidence in the images, such as broken parts or error messages, to suggest that the devices are faulty.
Potential Risk of Failure
No negative conditions such as severe wear, rust, or crushed cables have been observed in the images. The condition of the products appears to be good.