Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 17.11.2025
Löwenstein VentiLogic LS Respiratory Device Analysis Report
General Assessment and Product Description
The visuals show a medical respiratory device (ventilator) and its accessories presented within a carrying case. The device is designed to support patients’ breathing process or to completely take over respiration. The logos, labels, CE mark, and overall workmanship quality on the product give the impression that the device is an original product.
Brand and Model Information
As a result of detailed examinations, the brand and model of the device have been clearly identified.
- Brand: Löwenstein
- Model: VentiLogic LS
This information is confirmed by the “VENTIlogic LS” and “Löwenstein medical” inscriptions on the front of the device, and the “Löwenstein Technology GmbH” manufacturer information on the rear label.
Quantity and Package Contents
The visuals show a total of 1 unit of the respiratory device and its accompanying accessories. The package contents are listed below:
- 1 unit Löwenstein VentiLogic LS Respiratory Device
- 1 unit respiratory circuit tubing compatible with the device
- 1 unit power cable integrated with the device
- 1 unit black, protective carrying case
Condition and General State
The device generally appears used but in good condition. The front panel, screen, and keypad are clean. On the rear panel, particularly around the ventilation grille and the power cable inlet, there is light dust and dirt. It is understood that the device has been generally well-maintained and carefully used.
Physical Deformation Analysis
The physical condition of the product has been examined in detail. There are no cracks, breaks, or significant dents on the device’s casing or screen. There is only a slightly yellowed old tape residue on and near the rear label. Apart from these, no significant deformation impairing the device’s aesthetics has been observed.
Mechanical and Electronic Components
The mechanical and electronic components of the device have been visually assessed.
- Mechanical Components: All mechanical parts such as the device’s carrying handle, control knob, buttons, respiratory circuit outlet, and the filter cover on the rear panel are intact and in place.
- Electronic Components: The device’s screen is operational, and the image quality is clear. The power cable is connected to the device and shows no signs of crushing or wear.
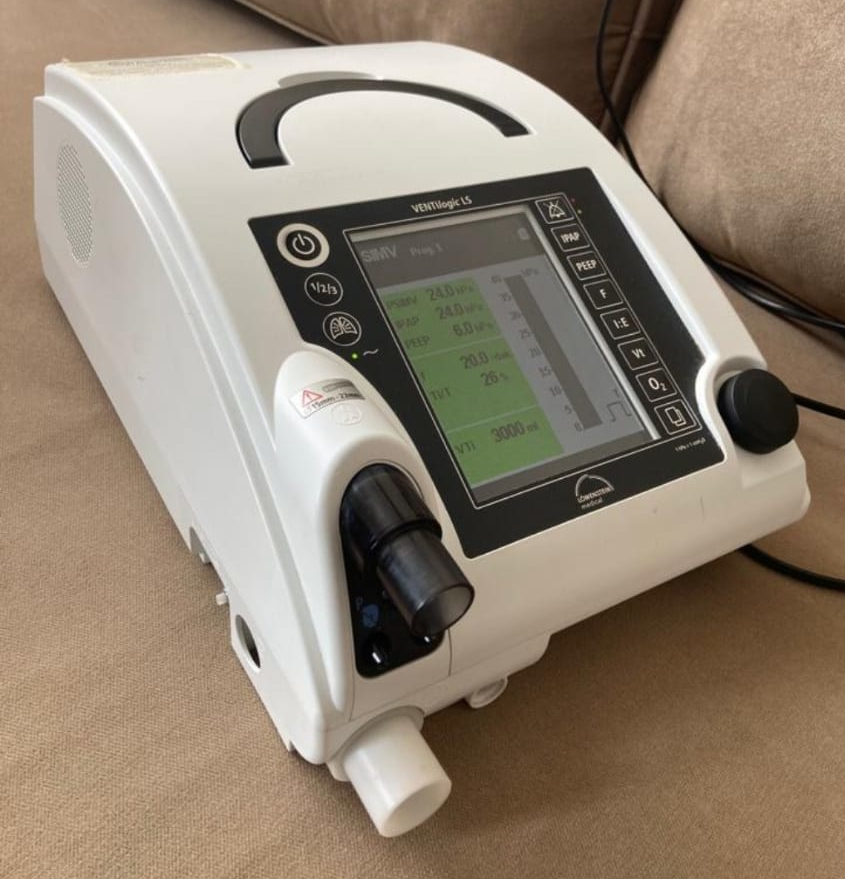
Screen Analysis and Operating Information
In one of the visuals, the device’s screen is active. The data displayed on the screen indicates that the device is operational and in “SIMV” mode. Some important parameters on the screen are as follows:
- Mode: SIMV (Synchronized Intermittent Mandatory Ventilation)
- Program: Prog 1
- Circuit Type: Single-limb vol.t
- PSIMV: 24.0 hPa
- IPAP: 24.0 hPa
- PEEP: 6.0 hPa
- Frequency (f): 23.5 /min. (breaths per minute)
- VTi: 2833 ml
There are no error codes or warning messages on the screen, which indicates that the device is currently operating stably.
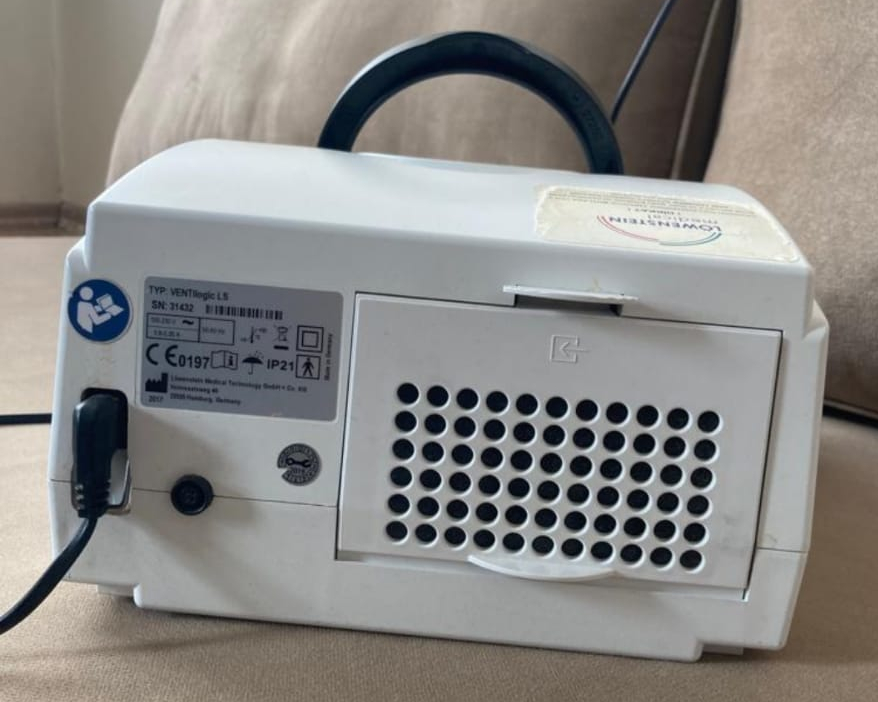
Label Information and Technical Details
The following information can be read from the product label located on the device’s rear panel:
- Type (TYP): VENTIlogic LS
- Serial Number (SN): 91432
- Year of Manufacture: The year 2017 is indicated within the box on the label.
- Certification: CE 0197
- Protection Class: IP21
The label also contains various symbols indicating the device’s compliance with medical device standards.
Condition of Accessories
The condition of the accessories accompanying the device is as follows:
- Carrying Case: The black colored case is specially designed for the device. Its internal compartments protect the device and its accessories in an organized manner. There are no visible tears or damage on the case.
- Respiratory Circuit: The tubing appears clean, but whether it is sterile or its usage status (new/used) cannot be determined from the visuals.
Potential Fault Risk and Conclusion
During the examination based on the visuals, no critical risk factors such as severe wear, corrosion, crushed cables, or broken parts that would directly affect the device’s operation have been detected. The overall condition of the device gives a positive impression that it can operate smoothly for a long time if regularly maintained and carefully used. In its current state, it shows no signs of malfunction.