Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 27.09.2025
Löwenstein VENTIlogic LS Respiratory Device Analysis Report
Overview and Product Description
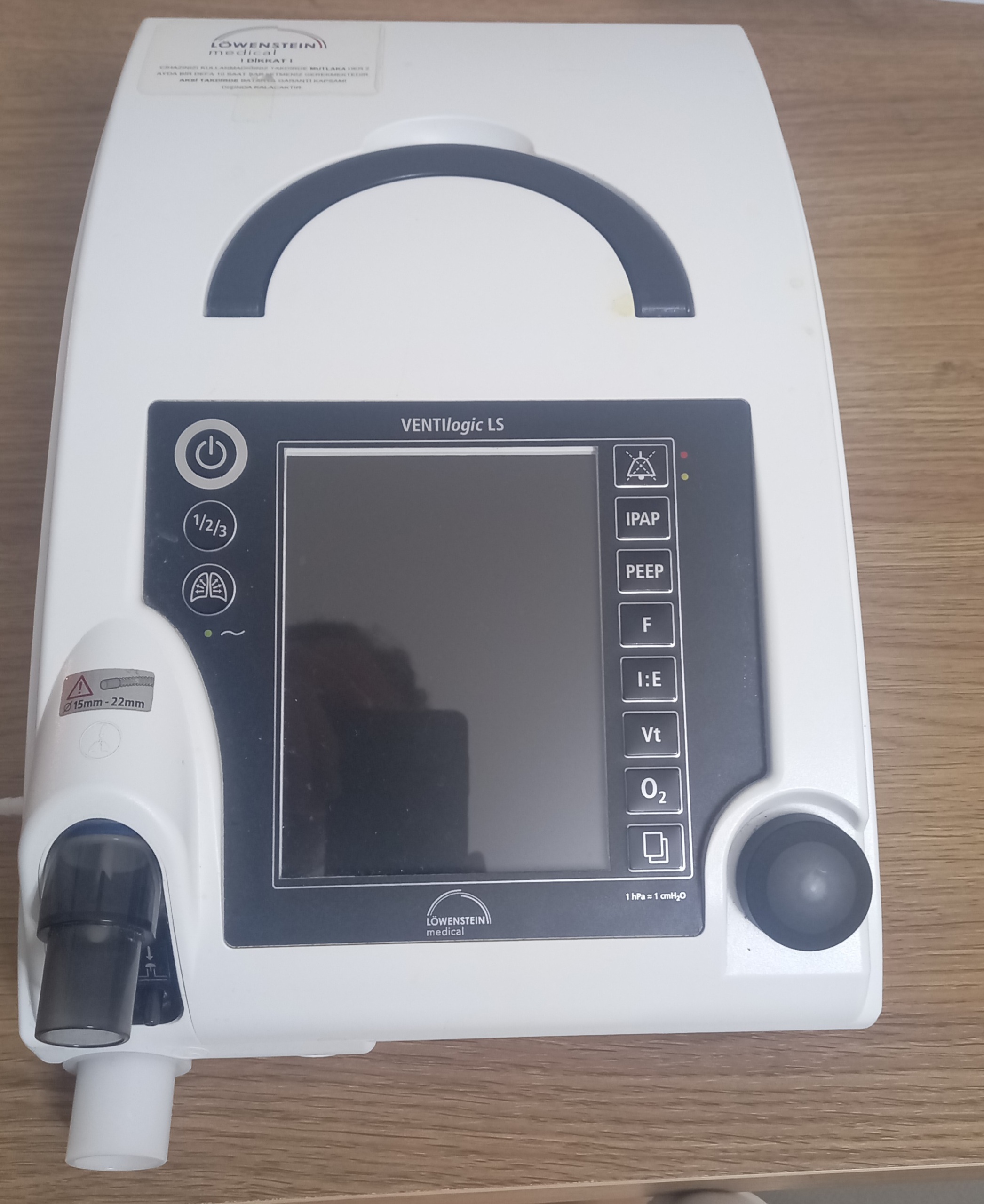
The device shown in the visuals is a ventilator used to provide respiratory support. The brand and model information on the product’s front panel is clearly legible. The device is generally known to be suitable for use in both adult and pediatric patients, whether in a home or hospital environment.
- Brand: Löwenstein Medical
- Model: VENTIlogic LS
Condition Assessment
Despite being used, the device appears to be in generally good condition. No major deformations such as severe impacts, cracks, or fractures were observed on its casing. The keypad and screen surface are physically intact. A general cleaning of the product would further enhance its aesthetic appearance.
Physical Examination
A detailed examination of the device’s white plastic casing revealed some signs of use. Specifically, there are slight yellowing marks on the right side of the upper carrying handle and on the top right corner of the casing. Some dust accumulation is present around the control knob and the screen. Since the device’s screen is off, it is not clearly understood whether it is operational or if there are any scratches on it; however, the screen appears to be free from physical damage.
- Casing: The white-colored casing has no major scratches or cracks. Only slight yellow stains and dust are present.
- Buttons and Control Knob: The power button, function keys, and rotary control knob are physically in place and appear intact.
- Connection Port: The port located at the lower left, where the patient circuit is connected, is clean and undamaged.
Technical Details and Label Information
Labels and inscriptions on the device provide information about some of the product’s technical specifications. However, detailed information such as serial number or manufacturing date could not be accessed.
- Warning Label: On the upper part of the device, there is a label written in Turkish. This label states that the device’s battery is outside the scope of warranty and needs to be charged for 10 hours once a month.
- Compatibility Information: Next to the connection port on the lower left side of the device, there is a label indicating compatibility with 15mm and 22mm diameter hoses.
- Function Buttons: The buttons next to the screen point to respiratory parameters such as IPAP, PEEP, F, I:E, Vt, O2. This indicates that the device has advanced ventilation modes.
Accessories and Battery
In the visual, no external accessories (such as power cable, patient hose, carrying case, etc.) other than the device itself are present. The phrase “AKÜ TAKIMIDIR” (Battery Pack) on the label confirms that the device has an internal battery. However, there is no visual evidence regarding the battery’s current performance and health status.
Potential Risk Assessment
Based on visual inspections, the device gives the impression of being well-maintained. The casing integrity is intact, and no significant wear or damage has been detected in its mechanical parts. Since there are no visually apparent serious physical flaws, there is no clear indication that the device, in its current state, poses a potential risk of malfunction.