Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 11.03.2026
Report code: 1773217217
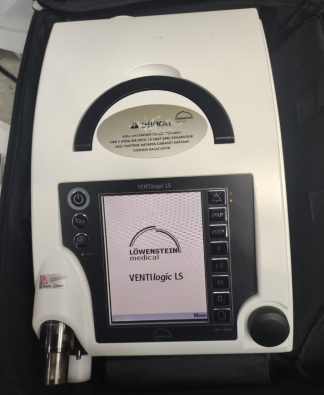
Löwenstein Medical VENTIlogic LS Mechanical Ventilator Analysis Report
Device Identification and Areas of Use
The product shown in the images is a mechanical ventilator device used to provide respiratory support to patients. Control buttons such as IPAP, PEEP, I:E, Vt, and O2 on its front panel indicate that the device has advanced respiratory modes. Thanks to the oxygen inlet port located on the left side of the device, it is suitable for use with external oxygen support. It is stated that the patient circuit outlet on the front is compatible with respiratory circuits with a diameter of 15mm-22mm. It has a design suitable for both home and clinical use.
Brand and Model Information
According to the information on the manufacturer’s label on the front panel and the rear of the device, its brand has been identified as Löwenstein Medical, and its model as VENTIlogic LS.
Authenticity Assessment
Upon examining the device’s casing craftsmanship, screen placement, keypad quality, manufacturer’s label on the rear panel, CE certification mark, and place of manufacture indication, the product is assessed to be original.
Quantity Information and Accessories
The images show 1 main device. No external accessories such as a power cable, respiratory circuit, mask, or carrying bag belonging to the device are present in the images.
General Condition and Physical State
The device is used and its general condition is good. There is slight dust accumulation and superficial stains on the casing due to use. No breaks, cracks, or deep scratches have been observed on the device’s outer casing, screen, or connection points.
Mechanical and Electronic Components
The black carrying handle located on the top of the device is intact. The membrane keypad on the front panel and the adjustment knob in the lower right corner are physically undamaged. The filter cover on the rear and the port covers on the sides are in place. The large digital screen on the front panel maintains its physical integrity. Since the device is in the off position, there is no data available regarding its electronic functions and screen menus.
Connection Ports
The connection points identified on the device are as follows:
- Front panel: 15mm-22mm patient circuit outlet port.
- Right side panel: Two data/communication ports and one round port with a rubber cover.
- Left side panel: Oxygen inlet port.
- Rear panel: Power cable input and one round connection socket.
Label Information and Technical Details
The information on the manufacturer’s label located on the rear of the device is as follows:
- TYP: VENTIlogic LS
- SN: 47601
- Power Requirement: 100-230 V, 50-60 Hz, 0.8-0.35 A
- Manufacturing Date: 2020-08
- Manufacturer: Löwenstein Medical Technology GmbH + Co. KG (Hamburg, Germany)
- Protection Class: IP21
On the left side panel of the device, there is a warning for oxygen input: O2 Max 15L/min <1000hPa. On the top of the device, there is an informative label prepared in Turkish, containing information about maintenance period, filter replacement period, and battery usage conditions. Additionally, a round service control label indicating the year 2022 is present on the rear panel.
Battery Status
The presence of warnings regarding battery usage conditions on the Turkish label on the top of the device indicates that the device has an internal battery. Since the device is off, there is no visual data regarding the battery’s current capacity or status.
Existing Faults and Potential Risks
No broken parts, missing hardware, or physical deformations that could impede the device’s operation have been detected in the images. The device’s external condition is quite good, and no significant potential fault risks have been observed based on visual data.