Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 25.10.2025
Löwenstein VENTILogic LS Ventilator Analysis Report
Overview and Product Description
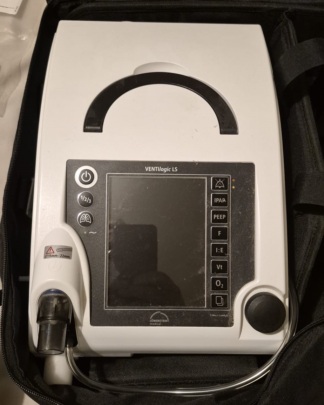
The product shown in the images is a mechanical ventilator device used to provide respiratory support. The device is designed for both invasive and non-invasive ventilation in hospital and home care settings. The screen and control buttons on the front panel allow for the adjustment and monitoring of the device’s ventilation parameters. Its portable nature is evident from the carrying handle located on its upper part.
Brand and Model Information
The brand of the device in the image is clearly readable as Löwenstein at the bottom of the screen. The model information is indicated as VENTILogic LS just above the screen. The device is a life support ventilator manufactured by Löwenstein Medical.
Physical Condition and Deformation Assessment
The device is generally in a used condition. Upon examining its physical state, the following details were observed:
- Casing: The device’s white plastic casing generally has a clean appearance. However, there is a slight yellowing or stain next to the black rotary knob on the lower right corner. Other than this, no significant cracks, fractures, or dents were detected on the casing.
- Screen: The device’s screen is off. Due to light reflection, some fingerprints and minor superficial scratches are visible on the screen surface. There are no cracks or deep scratches on the screen.
- Buttons and Controls: The power button, function buttons, and rotary control knob on the front panel are physically in place, and their symbols are clear. No wear or damage has been observed.
Technical and Hardware Details
The visual evidence regarding the device’s technical specifications includes:
- Patient Circuit Connection: A port for connecting the patient circuit is located on the lower left side of the device. The label next to this port shows 15mm and 22mm markings, indicating that the device is compatible with hose sets of these two different diameters.
- Power Connection: A black power cable emerges from the back of the device.
- Battery: It is understood from the instructions under the heading “BATARYA KULLANIM ŞARTLARI” (BATTERY USAGE CONDITIONS) on the top label that the device has an internal battery for portable use. There is no visual evidence regarding the current health or charge level of the battery.
Accessories and Missing Parts
Based on the visual inspection, the accessories provided with the device and the missing parts are as follows:
- Available Accessories: No accessories (carrying case, user manual, etc.) are visible other than one black power cable connected to the device.
- Missing Parts: Essential parts for the device’s operation, such as the patient circuit (breathing tube) and mask, are not present in the images.
Label and Warning Information
There are multiple informative labels on the device:
- Compatibility Label: At the patient circuit outlet, there is a small label indicating compatibility with 15mm and 22mm tube diameters.
- Usage Instructions Label: On the upper part of the device, there is a usage label prepared in Turkish. This label contains instructions regarding filter maintenance and battery usage. According to the label, the filter should be cleaned weekly or as needed and replaced every 6 months. For battery health, it is stated that the device should be discharged and recharged at least once every 10-12 months.
- Service Label: There is another label on the device with contact information for a company named SAPIO.
Information such as the device’s serial number, lot number, or REF code is not clearly legible in the images.
Potential Risk Assessment
The physical condition of the device is generally good, showing no significant signs of damage or malfunction. No serious risks such as cracks, dents, or cable wear have been observed. However, given that the device is second-hand, the lifespan and performance of the internal battery may be uncertain. Essential components such as the patient circuit and mask, which are missing, need to be procured. It cannot be determined from the images whether the device is functionally operational, and a technical inspection is strongly recommended before use.