Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 23.09.2025
Philips Respironics Trilogy 100 Ventilator Analysis Report
Overview and Product Description
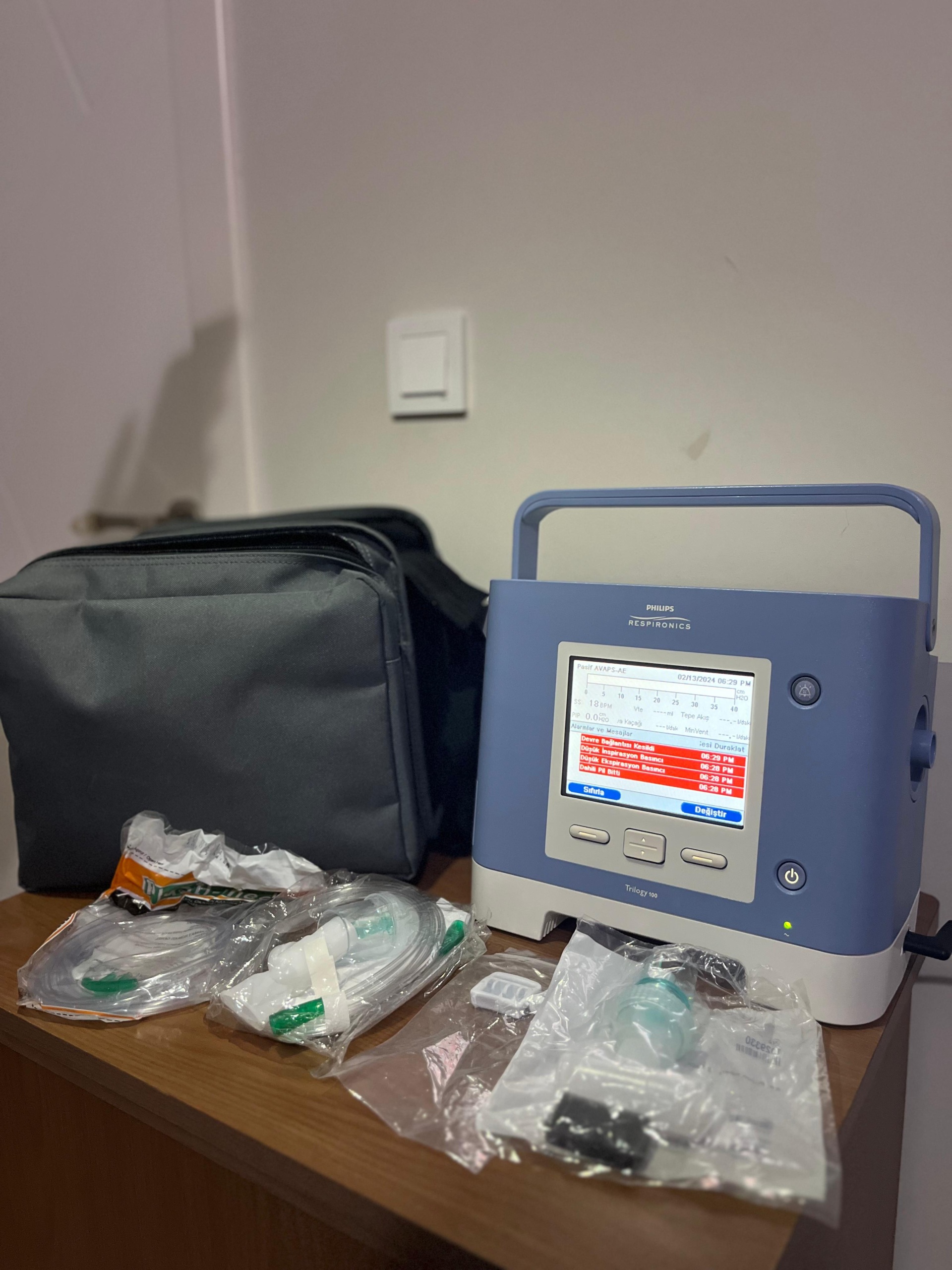
The product shown in the images is a ventilator and its accompanying accessories. The device is a ventilator designed for medical use, employed for patient respiratory support. Alongside the product, there is a carrying bag and various new, packaged medical consumables. The general condition of the device appears used but physically in good condition.
Brand and Model Identification
During the detailed visual inspection, the Philips Respironics brand and Trilogy 100 model name are clearly visible on the device’s front panel. This information definitively confirms the device’s identity.
Physical Condition and Deformation Analysis
The outer casing of the device generally has a clean and well-maintained appearance. Inspections revealed no significant deformation such as deep scratches, dents, cracks, or discoloration on the product’s surface. Mechanical components such as the device’s handle, buttons, and port entries are visually in good condition. The accessories appear to be in their original packaging and unused.
Technical and Electronic Evaluation
The electronic components of the device are in working order. The images show that the device’s screen is on and the power indicator light is green. However, an active alarm state is present on the device. Connection points such as the power input, USB port, and SD card slot are located on the rear panel of the device, and no physical damage has been observed on these ports.
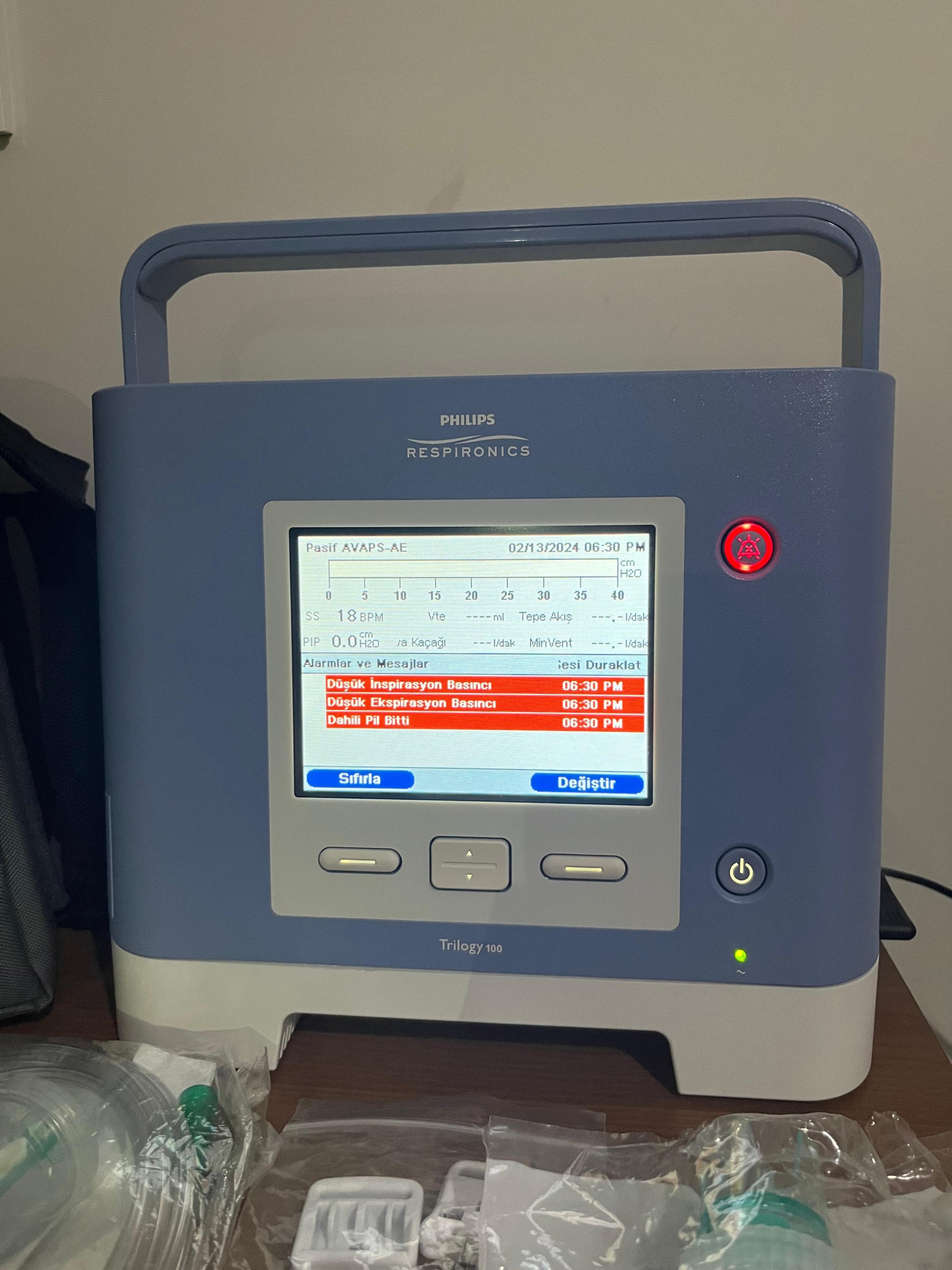
Screen Analysis and Current Status
The device’s screen is active and displays important information. The findings from the screen analysis are listed below:
- Date and Time: The screen displays “02/13/2024 06:30 PM”.
- Active Alarms: The device’s screen shows red warnings and a flashing red alarm light. The warning messages displayed on the screen are as follows:
- Low Expiration Pressure: This warning indicates that the pressure in the device’s breathing circuit is low. This situation could be related to connection issues or device settings.
- Internal Battery Depleted: This message clearly indicates that the device’s internal battery is completely discharged or that its lifespan may have ended.
Accessories and Package Contents
The following items are included with the device in the images:
- 1 unit Philips Respironics Trilogy 100 main unit
- 1 grey original carrying bag
- Multiple, new and unused various respiratory circuit parts, filters, and connectors packaged in clear plastic bags
The device’s power cable or adapter is not visible in the images, but the fact that the device is operational indicates it is connected to a power source.
Conclusion and Potential Risks
Although the device is physically in good condition, the “Internal Battery Depleted” warning displayed on the screen is a significant finding. This indicates that the device will not operate during power outages or in mobile use. The internal battery needs to be checked and likely replaced. The “Low Expiration Pressure” alarm highlights the importance of having the device inspected, calibrated, and its settings adjusted by an authorized technician before use. Safe use of the device may not be possible until the current alarms are addressed. Apart from these, no serious wear and tear, such as rusting or crushed cables, which could pose a potential risk of malfunction, has been observed on the device.