Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 23.04.2026
Report code: 1776939545
Mek MV2000 EVO5 Intensive Care Ventilator Analysis Report
Device Identification
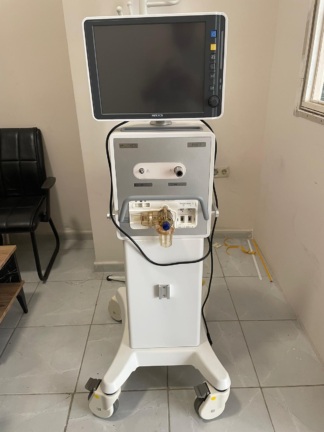
The images show a medical respiratory device (ventilator) designed for use in intensive care units, along with sealed packaging boxes and a promotional brochure that appear to belong to this device.
Brand and Model
The information on the promotional brochure and packaging boxes is clearly legible. The brand of the device has been identified as Mek, and the model as MV2000 EVO5. In the brochure, the product is referred to as the “MV2000 EVO5 ICU Ventilator.”
Usage Areas
According to the information in the brochure, the device is a high-performance intensive care ventilator. Its key features are as follows:
- Leak compensation with trigger synchronization that eliminates unnecessary patient discomfort and maximizes treatment effectiveness.
- Easy upgrade of the required treatment mode without changing the product, thanks to software-based product control.
- High Frequency ventilation mode providing care for children and patients with significantly reduced lung function.
- Remote monitoring of the patient’s condition in connection with a Central Monitoring System.
- Convenient product maintenance via Smart Viewer.
- Closed Suction support function that provides convenience for healthcare personnel and reduces the possibility of additional patient infection during aspiration.
Quantity Information
A total of 6 sealed cardboard boxes have been counted in the images. Of these boxes, 3 are large and 3 are smaller in size. Additionally, there is 1 promotional brochure. The contents of the boxes (the device itself, its stand, or accessories) could not be determined from the images as they are sealed.
General Condition and Physical Deformation
Since the device itself is not seen unpacked, no direct comment can be made regarding its condition. However, the general condition of the packaging boxes is good. The boxes are sealed with original yellow tapes and are placed on a pallet. No tears, dents, water damage, or serious deformation have been observed on the boxes. There are some handwritten notes and drawings on the promotional brochure.
Mechanical and Electronic Components
As the device is boxed, its mechanical and electronic components (screen, buttons, wheels, internal circuits, etc.) could not be visually analyzed. The representative image on the brochure shows that the device has a modern design with a touchscreen and a wheeled stand.
Accessories
Since the boxes are sealed, the accessories that come with the device (breathing circuits, humidifier, sensors, etc.) could not be identified. The brochure image shows the device’s stand and screen.
Label Information
There are labels on the packaging boxes; however, specific information such as serial numbers, lot numbers, or REF codes on these labels cannot be clearly read from the images. The model name “MV2000” and the phrase “INTENSIVE CARE VENTILATOR” are legible on the boxes. The “CE” mark is present on the brochure.
Documents and Year of Manufacture
No documents such as invoices, warranty certificates, or user manuals are visible in the images. There is no readable information on the labels regarding the manufacturing year of the device.
Potential Risk of Failure
Since the products are in their original sealed packaging and no signs of damage are found on the boxes, the risk of mechanical damage due to transport or storage appears to be low. It can be stated that the condition of the device is good.