Ready For Sale
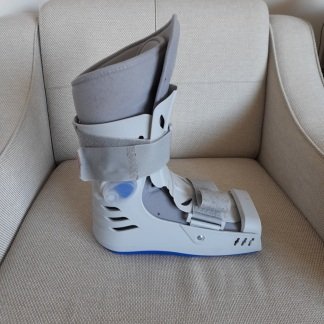
Secondhand Oped Vacoped Achilles Boot Foot Orthosis
Price: USD$ 120,00 Approx: 5.400,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 02.03.2026
OPED VACOped Achilles and Fracture Orthosis Boot Analysis Report
Device Description and Brand Information
The medical product in the examined images is an advanced orthosis designed for lower extremity (foot and ankle) stabilization. Based on the structural design, color codes (anthracite grey body, blue/turquoise adjustment points) shown in the images, and the documentation on the accessories, the device has been identified as the VACOped model belonging to the OPED brand. The brand name is embossed/printed on the straps. Additionally, the phrases “VACOped / VACOcast” are clearly visible on the information sheet inside the accessory bag.
Product Configuration and Part Count
The set shown in the images consists of the following components in total:
- 1 Main Orthosis Frame (Including outer shell, hinged structure, and strap system).
- 1 Vacuum-Cushioned Inner Liner (White terry cloth covered unit, either installed inside the boot or placed alongside it).
- 1 Insole/Wedge (Wedge-shaped, grey plastic part, marked “Medium”).
- 1 Accessory Package (Containing user manual/information sheet and 5 seals/clips in a sealed transparent bag).
Size and Fit Information
The word “Medium” is molded on the bottom surface of the detachable sole part (image 2). Based on this data, it is confirmed that the product is “Medium” size (Generally corresponds to shoe sizes 39-43). The product is designed for adult use.
Areas of Use and Indications
This orthosis is a functional stabilization boot utilizing “Vacuum Splint” technology. It is used in the following conditions:
- Conservative and post-operative treatment of Achilles tendon rupture.
- Stable foot and ankle fractures.
- Stabilization after ligament injuries.
- Severe sprains and post-operative rehabilitation process.
Mechanical and Physical Condition Analysis
Outer Shell (Casing): No deep cracks, fractures, or damage compromising the integrity of the rigid plastic (polyethylene/polypropylene derivative) skeletal structure have been observed. Slight superficial scratches due to use may be present on the plastic surfaces, but the overall structure is sound.
Hinge and Locking System: The blue caps and locks on the range of motion (ROM) adjustment mechanism located at the ankle level are in place. No signs of corrosion or rust are present.
Straps and Connections: Velcro fastening straps are present. The fabric texture of the straps retains its integrity; no tearing or excessive fraying is observed.
Inner Liner: The cleanliness of the white terry cloth surfaced inner liner is at an “acceptable” level according to visual inspection. No significant stains, tears, or discoloration are apparent.
Sole Structure: The rocker sole form is designed to support walking dynamics. No excessive wear or detached parts are visible on the sole rubber.
Label and Document Analysis
The following technical data can be read on the paper inside the accessory package provided with the product:
- Document Date Code: 2014 KW35. (This code indicates the 35th week of 2014. The manufacturing year might be this date or shortly thereafter).
- Content Information: The phrase “5 Siegel / 5 seals / 5 cachets” confirms the presence of 5 security seals (small green/blue pieces) used to secure the adjustment mechanism inside the package.
Accessories and Spare Parts Status
The device is observed with the essential accessories required for the treatment process. Specifically, the presence of the “seals,” used to prevent or monitor changes in angle adjustments by the patient, in their original package indicates that the product is a complete set. There is no external charger or electronic component; the system operates on mechanical and pneumatic principles.
General Assessment and Potential Risk Analysis
As a result of visual inspection, the device is in mechanically sound and functional condition. The 2014 document reference reveals the product’s age, but plastic and metal components have a long shelf life. Potential risks to consider include the possibility of the granules inside the vacuum cushion (liner) losing their properties over time or air leakage (which can only be determined by physical testing), and the reduction in the adhesion strength of the Velcro straps. However, visually, no damage requiring immediate intervention has been detected.