Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 13.07.2025
DeVilbiss 525KS Oxygen Concentrator Analysis Report
Overview and Device Description
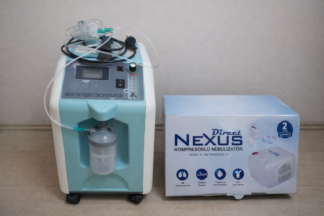
The product shown in the images is a DeVilbiss brand, model 525KS oxygen concentrator. The device is used to separate nitrogen from ambient air to produce high-concentration oxygen and deliver it to patients requiring respiratory support. It is understood that the product is designed for use in a home or clinical setting. The authenticity of the device is confirmed by the logo, label quality, and general workmanship details.
The total number of products analyzed in the report is as follows:
- 1 unit of DeVilbiss 525KS Oxygen Concentrator
- 1 unit of Water Humidifier Bottle
Physical State and Condition Assessment
The overall condition of the device is assessed as used. No significant color fading, cracks, or fractures were observed on its black plastic casing. There may be minor scratches or dust on the surface due to use, but no serious deformation has been identified from the photographs. The wheeled structure of the device enhances its mobility, and there is no visible damage to the wheels.
Mechanical Components and Controls
The mechanical parts on the front panel of the device have been visually inspected. The flowmeter, which adjusts the flow rate, appears to be physically intact, and its measurement lines are clear. There are no cracks or fractures on the panel containing the on/off switch and warning lights. The handle on the top of the device has a solid structure. No significant blockages or damage were observed in the ventilation grilles.
Accessories and Equipment
A transparent water humidifier bottle is seen attached to the device in the images. This bottle serves to humidify the oxygen delivered to the patient. Apart from the bottle, no other accessories (oxygen cannula, mask, power cord, etc.) are present in the images. Therefore, these parts should be considered missing. It is likely that a power cord input and other potential connection points are located on the back of the device, but these areas are not clearly visible in the images.
Technical Information and Label Details
The information on the rear label of the device is listed below. This label confirms the product’s technical specifications and identity.
- Brand: DeVilbiss Healthcare
- REF Code: 525KS
- Serial Number: There is a barcode on the label, but the numerical serial number underneath it is not clearly legible. Below the barcode, there is a code string: “525KS-693 REV C TAB B”.
- Electrical Ratings: 220 V – 230 V, 50 Hz, 1.55 A
- Classification: Class II Type B
- Standards and Certifications: CE 0044, IEC 60601-1, DIN ISO-8359
- Place of Manufacture: MADE IN USA of U.S. and Imported Parts
Potential Malfunction Risks and Conclusion
Based on the visual inspection, no critical risks that could lead to malfunction, such as severe wear, rust, or crushed cables on the casing or mechanical components, have been observed. The overall condition of the device appears to be good for its age and usage status. However, this report is based solely on a visual analysis and does not provide information about the condition of the internal components or the operational performance of the device. Consumables such as the humidifier bottle and filters may require periodic replacement. The stated potential risks are only a forecast based on visual inspection and do not imply that a definite malfunction will occur.