Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 04.10.2025
Plusmed Oxygen Concentrator Analysis Report
Product Overview
The device shown in the visuals is a Plusmed brand oxygen concentrator. Its primary function is to draw in ambient air, separate nitrogen, and provide concentrated oxygen to the user. It is understood to be used for medical respiratory therapies. This report covers the analysis of one main device and its attached accessories.
General Condition and State
The general condition of the device is assessed as used. General yellowing and discoloration are observed on its plastic casing, which may occur over time or due to exposure to sunlight. Apart from this, the device’s integrity is intact, and it shows no signs of major physical damage. Its appearance is consistent with the expectation of a regularly used medical device.
Physical and Mechanical Analysis
Upon detailed examination of the device’s outer casing, no significant cracks, breaks, or dents were detected. Only minor superficial scratches and general fading are present, attributable to its age and usage. The wheels located at the bottom are in place, and there appears to be no issue hindering its mobility. The flowmeter (flow gauge) that adjusts the oxygen flow and the humidifier reservoir are securely in place.
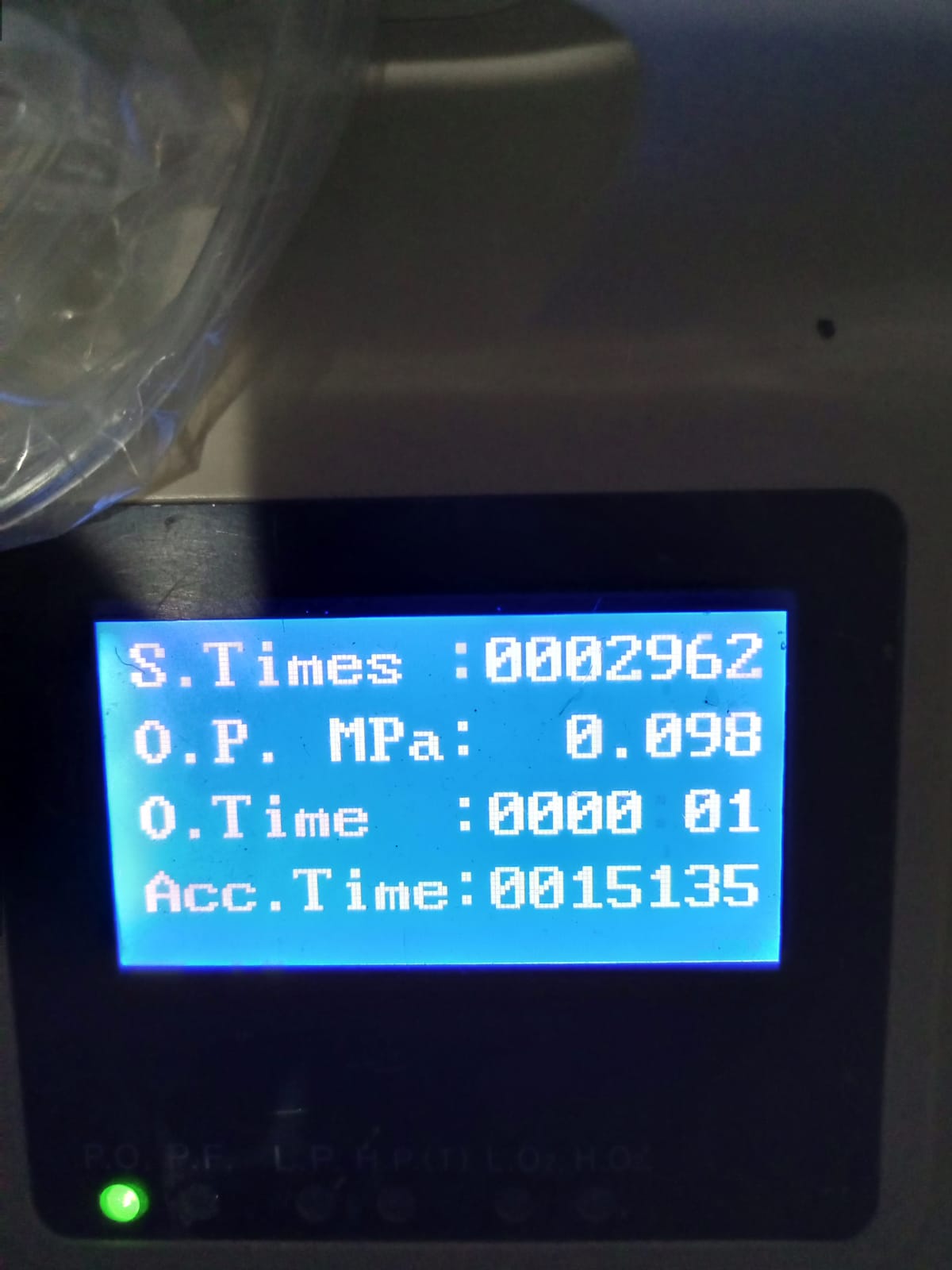
Electronic Components and Screen Analysis
The device’s electronic system is operational. One of the visuals shows the device’s screen active, displaying various operational data. The screen light is vivid, and no pixel loss has been observed. The information displayed on the screen is detailed below:
- S.Times : 0002962 – Indicates the number of times the device has been switched on and off, understood to have been operated 2962 times.
- O.P. MPa : 0.098 – Shows the device’s output pressure (Outlet Pressure).
- O.Time : 0000 01 – Indicates the duration of the current operating session.
- Acc.Time : 0015135 – Shows the device’s total operating time (Accumulated Time). This data reveals that the device has been used for a total of 15,135 hours to date.
A green status light, interpretable as “P.O.” (Power On), is illuminated below the screen.
Label Information and Warnings
On the front of the device, there is a label containing important warnings in both Turkish and English. The prominent information on this label includes:
- The user manual must be read before operating the machine.
- The dust filter should be changed once a month, and the air inlet filter once a year.
- Oxygen concentration is at 93%±3.
- Must not be brought near cigarettes, open flames, or substances like oil/grease.
A label containing the device’s serial number, REF code, or lot number cannot be clearly read in the visuals.
Accessories
The following accessories belonging to the device have been identified in the visuals:
- One humidifier (water) bottle attached to the device.
- A transparent tube (nasal cannula or intermediate connection tube) connected to the oxygen outlet.
- One black power cable coiled in the slot on the side of the device.
No other accessories such as a box, extra filters, or a user manual are visible.
Potential Malfunction Risk and Assessment
The total operating time of 15,135 hours displayed on the device’s screen indicates that the device has been used intensively. In oxygen concentrators, the molecular sieve beds that separate oxygen from air have a certain lifespan. This high operating hour count suggests a possibility of decreased efficiency and the need for maintenance or replacement of these components in the future. However, the current visuals show no evidence of any error codes, warning lights, or functional malfunctions related to the device. The device is in working condition and has no serious mechanical or physical flaws. Its general condition is normal, considering its usage history.