Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 11.12.2025
RespiroX 7F-3 Oxygen Concentrator Analysis Report
Device Identification and Information
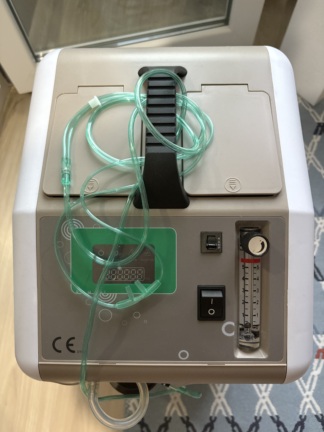
The product in the image is an oxygen concentrator, which falls into the category of medical respiratory support devices. Upon examining the labels and logos on the product, the brand was clearly identified as RespiroX, and the model as 7F-3. The phrase “Oxygen Concentrator” is present on the front face of the device, defining its function.
Areas of Use and Purpose
This device takes ambient air, separates nitrogen gas from it, and provides high-concentration oxygen to the user. It is used for home-based respiratory therapy for patients suffering from COPD, asthma, lung diseases, or low oxygen saturation. It is a stationary, electric-powered unit typically positioned at the patient’s bedside.
Quantity and Accessories
The image shows 1 unit of the main device. The components observed integrated with or attached to the device are:
- 1 Humidifier Bottle – Attached in the device’s middle compartment.
- 1 Power Cable – Partially visible on the floor, next to the device.
General Condition and Cosmetic Examination
The device is in used condition and is not new. Upon examining its general condition, the following details are noteworthy:
- Casing Cleanliness: Black stains, dirt accumulation, and scuff marks are present on the lower parts of the product’s white outer casing, especially on the bottom right and left corners.
- Signs of Wear: Slight fading or dirtiness may be observed on the surfaces where the front panel texts are located, but the brand and model inscriptions are legible.
- Middle Compartment: Dust/sediment accumulation has been observed inside and at the base of the transparent covered section where the humidifier bottle is located. It is understood that this area requires hygienic cleaning.
Technical Components and Controls
The technical analysis based on the image is as follows:
- Display: The LCD screen on the upper part of the device is active. The numbers “03450” are readable on the screen. In medical oxygen devices, such counters usually indicate the total running hours.
- Signal Lights: A green LED light is illuminated on the right side of the display panel. This indicates that the device has power connection and is on/operating or in standby mode.
- Flowmeter: A ball-type flowmeter is located vertically on the right side. Its adjustment knob is in place. The scaling, as far as can be discerned from the image, is in liters/minute (L/min).
- Oxygen Outlet: A metal outlet port for cannula connection is present on the left side of the device. No hose is attached to this part.
Battery and Power Status
This type of oxygen concentrators (such as the 7F-3 model) generally do not operate with an internal battery; they are connected to mains electricity (220V). The black cable visible on the floor in the image is the device’s power supply cable. The illuminated screen light of the device proves that it is connected to electricity at that moment.
Potential Malfunction Risk and Warnings
No major break, crack, or dent compromising the mechanical integrity of the device was detected during the visual inspection. However, the following points should be considered:
- Hygiene Status: The dirtiness on the lower parts of the device and in the humidifier reservoir suggests that the device has not been cleaned for some time. As it is a respiratory device, a detailed disinfection is required before use.
- Filter Condition: The device’s filters (usually located under a cover at the back or side) are not visible in the image, but considering the external dirtiness, it is highly probable that the internal filters are due for replacement.
Documents and Warranty
No invoice, warranty certificate, or label indicating an expiration date was found in the image. The operating hours on the device (around 3450 hours), while not providing definitive information about the warranty period, indicate that the device has been used intensively for a certain period.