Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 26.07.2025
Respirox Oxygen Concentrator Analysis Report
Overview and Device Identification
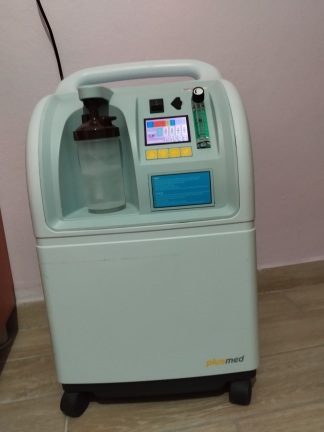
The product shown in the images is a medical device that produces high-purity oxygen by separating nitrogen from the ambient air. The device is labeled with the phrase “OXYGEN CONCENTRATOR”. The primary purpose of the product is to provide a continuous supply of oxygen to patients in need of respiratory support. Additionally, the “Nebulizing interface” outlet indicates that the device also has a nebulizer function, allowing for the inhalation of vaporized medication.
Brand and Model
The brand of the device in the images is Respirox, which is clearly visible on the front panel. Two different model codes are noticeable on the device: 7F-5B and SZ-5AW. This suggests that the images may belong to two very similar models from the same brand, or to different versions of a single model.
Quantity and Content Information
Upon examination of all the images, the following components have been identified:
- 1 Respirox Oxygen Concentrator main unit
- 1 transparent water humidifier bottle attached to the device
- 1 black power cord and plug connected to the device
External user accessories such as an oxygen cannula, mask, or nebulizer kit are not present in the images.
Physical State and Condition
The device has a generally clean and well-maintained appearance, giving the impression of being lightly used. No cracks, fractures, dents, or major scratches are observed on its plastic casing. Its slightly off-white color could be its original color or a minimal discoloration that has occurred over time. The overall visual condition of the device is good.
Mechanical and Electronic Components
The mechanical and electronic components of the device appear to be visually intact. The flow adjustment knob (Flowmeter), power switch, and other control buttons on the front panel are physically in place and do not appear damaged. The Oxygen Outlet and Nebulizing Interface ports are clean and solid. No crushing, peeling, or damage has been detected on the power cord and plug.
Screen Analysis and Usage Time
In the image showing the device in operation, the digital screen is seen working properly with a blue backlight. Next to the screen, the phrases “COUNTS (hours)” and “TIMING (minutes)” are present.
- Usage Time: The value “32” is readable on the screen, which can be associated with the “COUNTS (hours)” heading. This value indicates that the device has been used for a total of only 32 hours. This duration is quite low for an oxygen concentrator and signifies that the device has been used very little.
- The screen also displays values such as “140” and “0.00”. These are presumed to be related to session duration or other operational data.
Label and Technical Information
On the back of the device, there is a label with warnings in both Turkish and English. This label contains safety instructions regarding the correct use of the device. However, the label containing detailed technical information such as the device’s serial number, REF code, lot number, or year of manufacture is not clearly legible in the images.
Potential Malfunction Risk Assessment
Based on the visual inspection, no obvious potential risk of malfunction has been observed. The power cord is intact, there is no critical damage to the casing, and there is a screen image confirming the device is operational. The low total usage time of 32 hours is a positive indicator that the internal components of the device may not be excessively worn. It should be noted that this assessment is a forecast based solely on visual evidence, and the full functional status of the device can only be confirmed through a technical test.