Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 12.01.2026
Yuwell 8F-5A Oxygen Concentrator Analysis Report
Device Identification
The device in the examined visuals is a wheeled and portable medical oxygen concentrator used to provide respiratory support. The device is an electronic medical equipment that provides high-purity oxygen to the user by taking ambient air and separating it from nitrogen.
Brand and Model
As a result of detailed visual inspection of the product, brand and model information has been definitively identified:
- Brand: Yuwell
- Model: 8F-5A
This information is clearly legible on the front face of the device, below the LCD screen, and on the rear label. Furthermore, the label information includes “JIANGSU YUYUE MEDICAL EQUIPMENT & SUPPLY CO., LTD.” as the manufacturer.
Originality
The material quality of the device’s outer casing, the neatness of the screen assembly, the print quality of the “Yuwell” logo, and especially the details of the regulation label on the rear panel (which includes a QR code and serial numbers compliant with GS1 barcode standards) indicate that the product is an original Yuwell production.
Areas of Use
This device (model 8F-5A) is used in hospitals or home care services to meet the oxygen needs of patients suffering from COPD (Chronic Obstructive Pulmonary Disease), asthma, or other respiratory insufficiencies. It is a device capable of providing oxygen at a maximum flow rate of 5 Liters/Minute.
Quantity Information
The visuals show 1 main device and numerous installation accessories belonging to the device. The set contents are as follows:
- 1 Yuwell 8F-5A Oxygen Concentrator Main Unit
- 1 Water reservoir (Humidifier jar/beaker)
- 1 Humidifier connection tube
- 1 Oxygen cannula (in its bag)
- 2-3 spare filter felts (in their bags)
- 1 Remote Control
- User manuals and Warranty Certificate
General Condition and Physical Condition
The device’s outer casing is made of white hard plastic and is visually extremely clean. No yellowing, stains, or signs of use are observed on the casing. The condition of the wheels and the cleanliness of the bottom plastic components suggest that the device has had almost no floor contact or has only been tested for a very short period indoors. The front panel, buttons, and flow meter are spotlessly clean.
Physical Deformation
During the examination, no scratches, dents, cracks, breaks, or discoloration were detected on the casing. The water reservoir housing and air outlet ports are intact. The carrying handle is not deformed.
Screen Analysis and Usage Time
The LCD screen located on the top panel of the device has been examined in detail. The screen is functional, and the following values are legible:
- Displayed Value: 00004
- Meaning: This number, located under the “TIMING” and “COUNTS” labels, indicates the total operating time of the device. The value 00004 suggests that the device has been used for only 4 units (likely hours or cycles). This data proves that the device is in “Zero Condition” and was probably only turned on for testing purposes.
Mechanical Components
The “Flowrate” adjustment knob (column containing the flowmeter ball) located on the front panel appears sturdy. The four swivel wheels at the bottom are complete and assembled. The air filter cover (“Pull out to replace the filter”) on the rear panel is in place and maintains its mechanical integrity.
Electronic Components and Technical Data
The technical specifications indicated on the device’s rear label are as follows:
- Power Supply: 230V~, 50Hz
- Input Power: 350VA
- Oxygen Purity: 93% +/- 3% (at 5L/minute flow)
- Model: 8F-5A
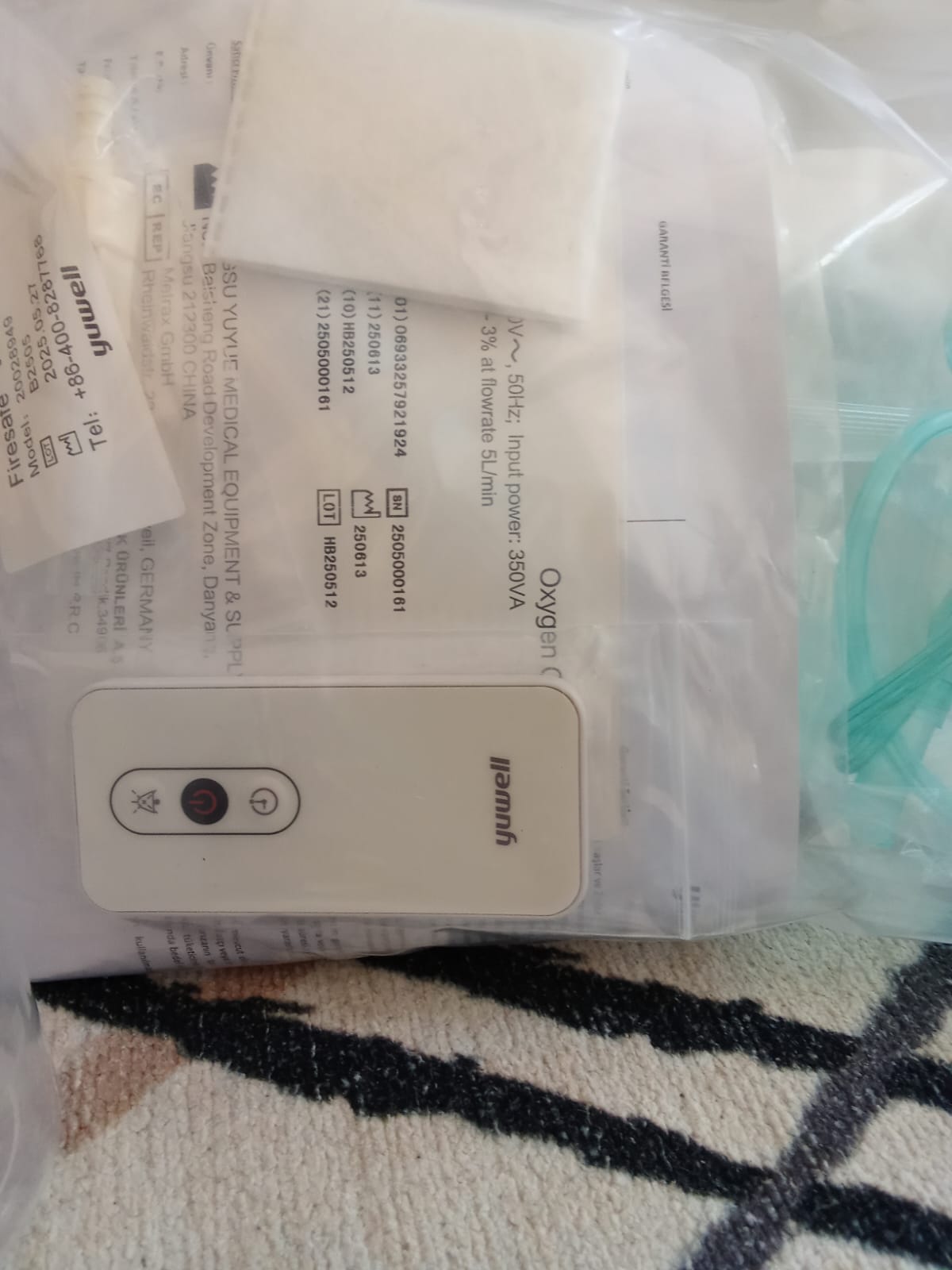
Accessories and Remote Control
The accessories provided with the device appear in their packaging:
- Humidifier Cup (Beaker): In a clean, limescale-free, and uncracked condition, ready to be mounted on the device. A “Safe Valve” warning is present on the lid.
- Remote Control: A white “Yuwell” branded remote control is present. Symbols for Power (On/Off), Timer, and Mute (or minus/plus) functions are visible on it.
- Cannulas and Filters: Appear in unopened hygienic bags.
Battery Status
Oxygen concentrators (excluding portable battery-powered models, like home-use devices such as this model) generally operate directly with mains electricity (plug). A thick, grey power cable emerges from the back of the device. There is no expectation of an internal battery in this model; it is designed to operate with mains electricity.
Label Information
The identifying codes on the silver label on the rear panel and in the documents are listed below:
- REF / GTIN: (01) 06933257921924
- Expiration / Production Date Code (11): 250613 (This code is usually in YYMMDD format, indicating June 13, 2025.)
- LOT Number (10): HB250512
- Serial Number (21): 2505000181
Production Year and Dates
On the label of the documents (warranty certificate/manual) seen alongside the device, the date “2025.05.27” is clearly printed. The (11) code on the device label indicates June 2025. This data confirms that the device was produced in mid-2025. The device is a relatively new production.
Documents
A “WARRANTY CERTIFICATE” with Turkish phrases is visible in the visuals. The presence of the document indicates that the product has been legally imported for the Turkish market or sold through a distributor. Additionally, security warnings with the phrase “Firesafe” and the heading “Oxygen Concentrator” are present on the paper.
Current Faults and Risks
During the visual inspection, no cracks, cable fraying, rust, or any “error” message were detected on the device. The “00004” counter value indicates that the device is not worn out. No mechanical or cosmetic fault risk is apparent.
Potential Fault Risk
Since the product appears extremely clean and new, no fault risk due to physical wear and tear is observed in the near future. Only ensuring the correct connections of the humidifier cup and timely replacement of filters are important for routine maintenance.