Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 24.08.2025
Inogen One G3 Oxygen Concentrator Analysis Report
Overview and Device Identification
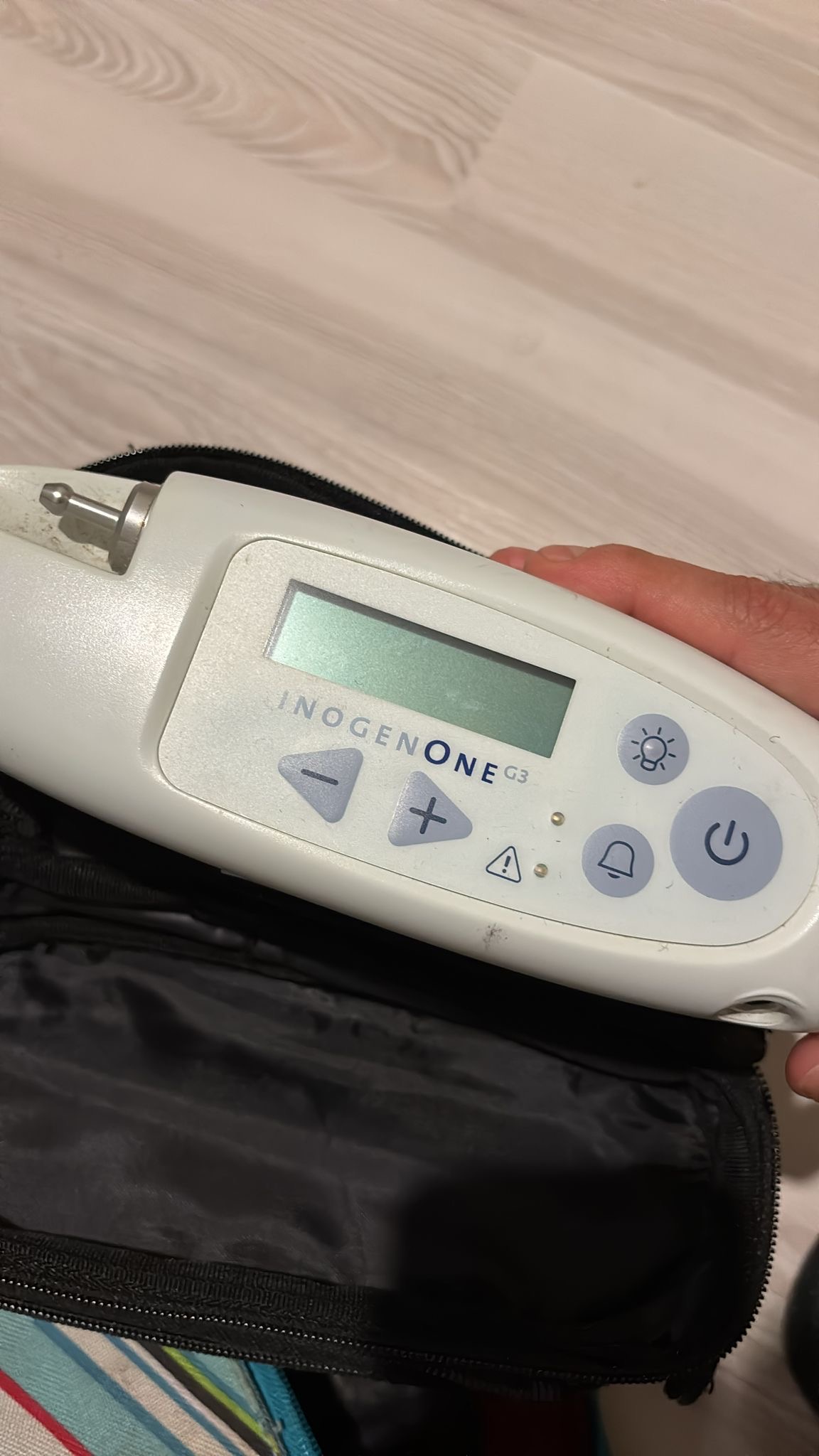
The visuals show one portable oxygen concentrator and a carrying bag understood to belong to this device. The brand and model of the device have been clearly identified as Inogen One G3 from the labels and inscriptions on it. The model number is stated as IO-300.
Area of Use and Originality
This product is a portable medical device used to provide a higher concentration of oxygen from ambient air to individuals requiring supplemental oxygen. Based on the logo, font characters, label quality, and overall workmanship details on the device, it is understood to be an original product.
Quantity Information
A total of 2 items are present in the visuals:
- 1 Inogen One G3 Oxygen Concentrator
- 1 black carrying bag
General State and Condition
The general state of the device is assessed as used. There are noticeable dirt and stains on its casing. Dust accumulations are observed, particularly around the control panel and on the lower ventilation grilles. The carrying bag also appears to have been actively used. However, its overall condition does not indicate a functional problem.
Physical and Mechanical Assessment
The physical and mechanical components of the product have been examined in detail:
- Casing: No major deformations such as deep scratches, breaks, or cracks were observed on the device’s light-colored plastic casing. However, minor scratches and general dirtiness are present in various areas.
- Buttons: The power, volume, illumination, and settings buttons on the control panel are physically in place and do not show any significant damage.
- Air Filters: The two air filter slots located at the bottom of the device are seen to have filters installed, and dust accumulation is present on them. These filters may need cleaning or replacement.
- Screen: The device’s LCD screen is in an off state. No breaks, cracks, or stains were detected on the screen.
Label Information and Technical Details
Important information is present on the label located at the bottom of the device. According to this information, the device is suitable for in-flight use as approved by the Federal Aviation Administration (FAA). The information on the label includes:
- Brand: Inogen
- Model: Inogen One G3 Oxygen Concentrator
- Model No: IO-300
- Serial Number (SN): 18279500
- Input: 13.5-19 V / 85 W
- Output: 90% Oxygen
- Manufacturer Information: INOGEN INC. 326 BOLLAY, GOLETA, CA 93117, USA
Additionally, the label includes various certification marks such as CE, cCSAus, and important safety warnings like “NO SMOKING” and “NO OIL OR GREASE”.
Accessories
The visuals show one black, mesh, and adjustable-strap carrying bag belonging to the device. The device is positioned inside the bag, and no tears or significant deformations were observed. No other accessories (power cable, battery, cannula, etc.) are present in the visuals.
Potential Malfunction Risk
Based on the evidence in the visuals, no obvious malfunction (broken part, error code, etc.) that would directly impede the device’s operation is observed. However, the dust and dirt accumulation on the air intake filters and casing indicates a need for maintenance and cleaning, as it could affect the device’s performance. It is anticipated that the device is in generally good condition and could operate without issues after maintenance.