Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 22.09.2025
Inogen Rove 6 Portable Oxygen Concentrator Analysis Report
Device Description and Intended Use
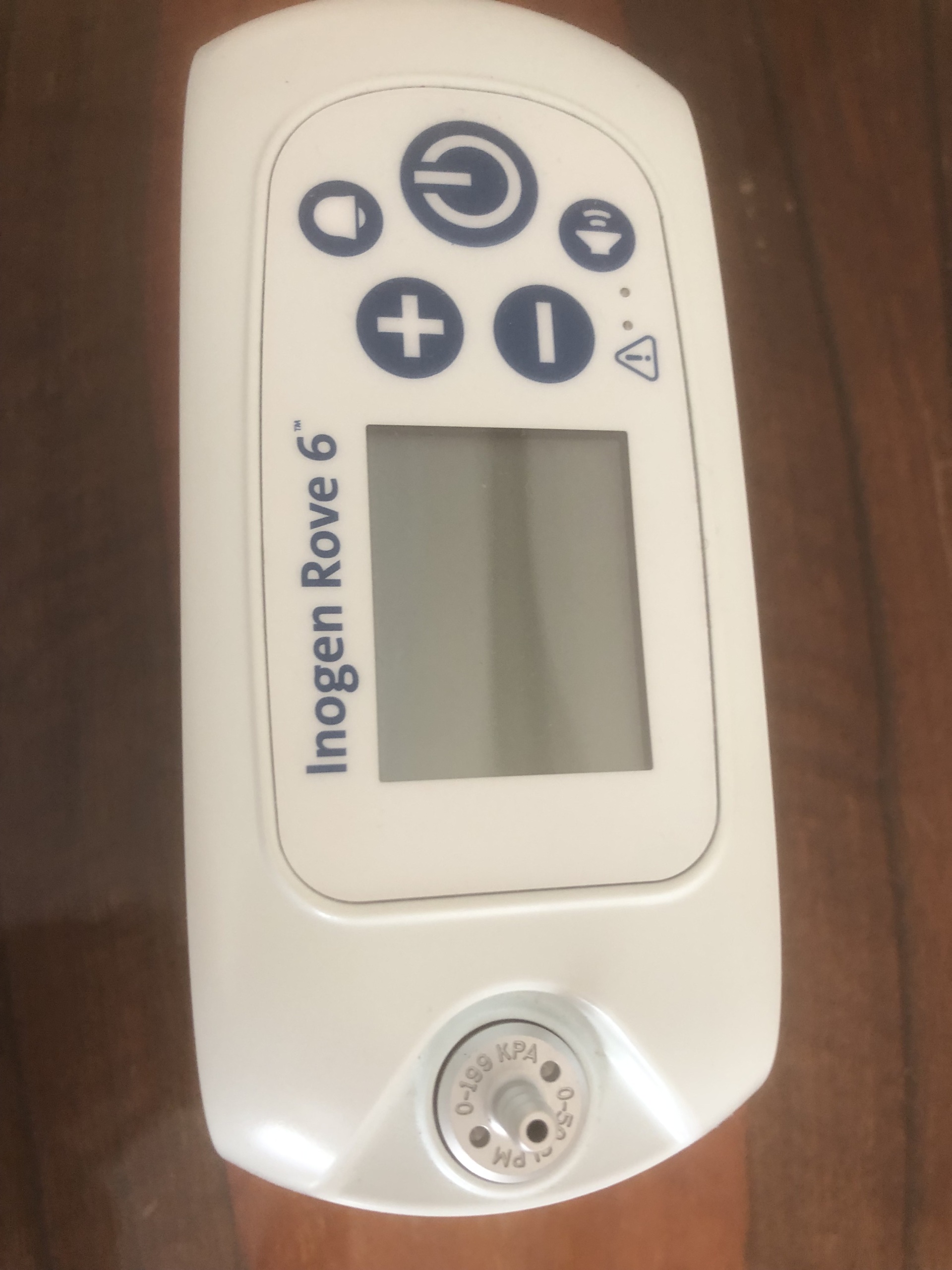
The visuals show an Inogen Rove 6 model portable oxygen concentrator. This device is designed to provide supplemental oxygen on a prescription basis to users requiring respiratory support. Its compact and lightweight design makes it suitable for use at home, outdoors, or while traveling, offering users mobility and freedom. The airplane symbol on the label at the back of the device indicates that it is approved for in-flight use by the FAA (Federal Aviation Administration).
Brand and Model Information
The brand and model of the device examined in the visuals have been clearly identified:
- Brand: Inogen
- Model: Rove 6
This information is clearly legible on both the front panel of the device and the technical information label on its back. The logos, fonts, and overall design indicate that the product is original.
Physical Condition and Deformation Assessment
Visually, the device appears to be in a new or very lightly used condition. Its overall condition is very high. The following details were observed during the physical inspection:
- Casing: No scratches, cracks, dents, or discoloration were observed on the device’s white plastic casing. Its surface is clean and spotless.
- Screen: The screen on the control panel is off, but there are no scratches or damage on its surface.
- Buttons: The power, increase/decrease setting, and other function buttons appear intact and unworn.
- Mechanical Components: The air inlet filter and grille on the side of the device are clean and undamaged. There is no deformation or rusting on the metal oxygen outlet port located at the top.
Technical Label Information
Important technical and regulatory information is located on the label on the back surface of the device. This information is listed below:
- Model: Inogen Rove 6 Portable Oxygen Concentrator
- Reference Code (REF): IO-501
- Serial Number (SN): N11166699
- UDI: (01)00840171105953(21)23040049-EU
- Power Input: 12.0 – 16.8 VDC, 10.0 A
- Power Consumption: 120 W
- Oxygen Output: 90% Oxygen
- FCC ID: N7G-BX31A
- IC: 2417C-BX31A
- Protection Class: IP22
The label also features various conformity marks such as CE, UKCA, MD (Medical Device), and Intertek certification marks. The manufacturer’s address is stated as “INOGEN INC., 301 COROMAR DRIVE, GOLETA, CA 93117, USA”.
Overall Assessment and Conclusion
Only one Inogen Rove 6 main unit is present in the visuals; accessories such as the box, charging adapter, carrying bag, or nasal cannula are not available. The device’s overall condition is near perfect, and there are no signs of malfunction (broken parts, error messages, etc.) on it. Due to its very well-preserved physical state, it does not pose any visible potential risk of malfunction. Since the product’s screen is off, information such as operating hours could not be accessed, and no documents (invoice, warranty) are present in the visuals.