Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 09.12.2025
Inogen Rove 6 Portable Oxygen Concentrator Analysis Report
Device Identification and Brand/Model Description
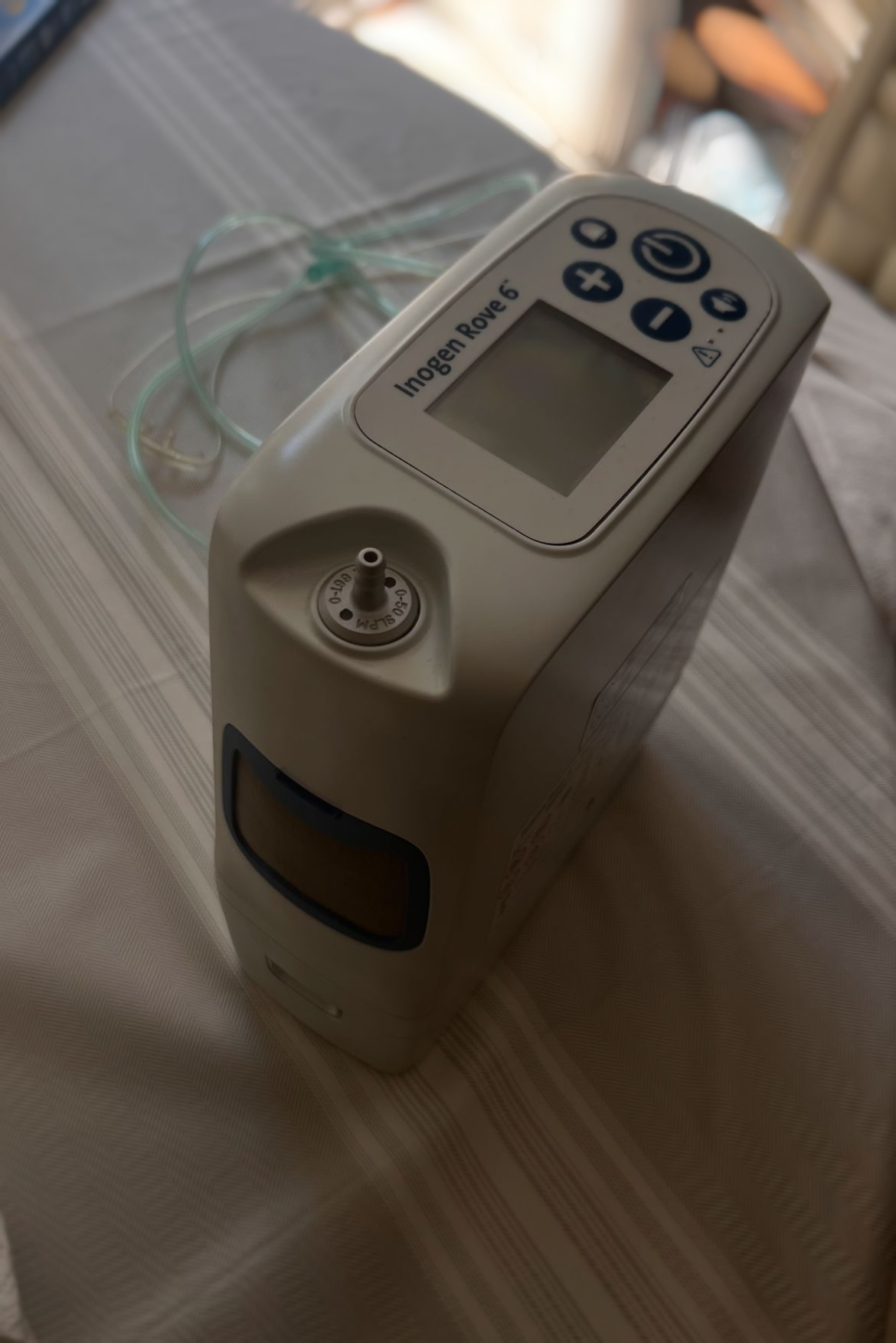
As a result of a detailed visual inspection, the device displayed has been identified as a portable oxygen concentrator used for medical purposes. The inscription “Inogen Rove 6” is clearly legible on the control panel and immediately above the digital screen located on the front of the device. Based on this visual evidence, the product’s brand has been determined as Inogen, and its model as Rove 6. Additionally, a large “Inogen” logo is present on the device’s original cardboard box.
Originality and Place of Manufacture
The product’s design language, button placement, screen structure, and material quality appear to be in line with Inogen brand standards. The brown cardboard box shown in the visuals features the brand’s logo and the inscription “Assembled in Czech Republic” at the bottom. The box, device, and carrying bag appear to be compatible with each other, and the product is evaluated as original.
Areas of Use
The Inogen Rove 6 is designed for patients suffering from respiratory difficulties, chronic lung conditions such as COPD, or those requiring supplemental oxygen therapy. Its portable design allows patients to receive oxygen outside the home, during travel, or during daily activities. The device takes ambient air, separates nitrogen, and delivers concentrated oxygen to the user.
Visible Accessories and Quantity Information
The following parts have been identified as a set in the visuals:
- 1 Inogen Rove 6 Oxygen Concentrator device
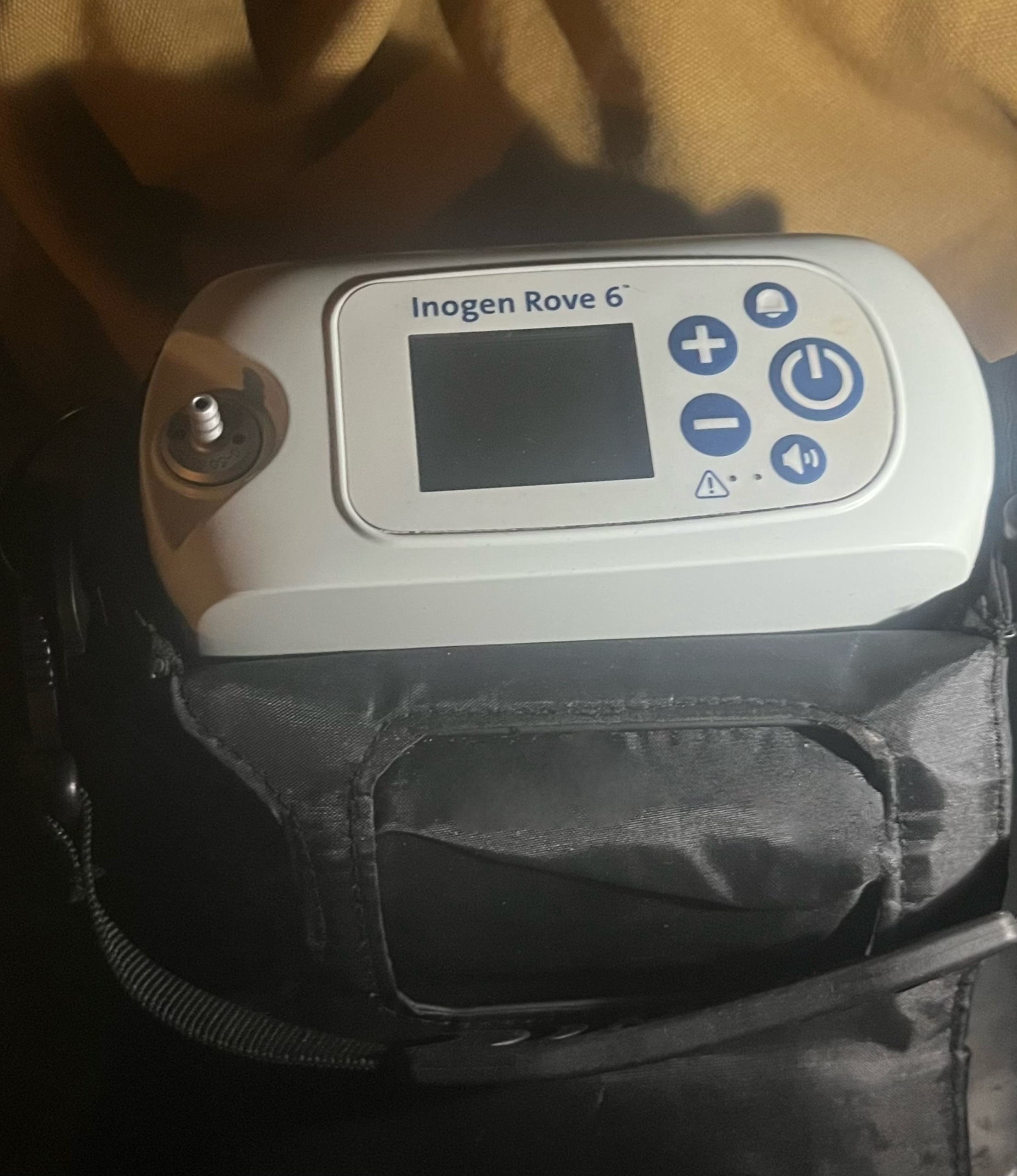
- 1 black shoulder-strap carrying bag fully compatible with the device
- 1 original “Inogen” branded cardboard box
- 1 silicone nasal cannula (oxygen tubing) suitable for patient use
General Condition and Physical State
The general appearance of the device gives the impression of being new or very lightly used. No yellowing, dirt, stains, or signs of wear have been observed on its white outer plastic casing. The protective bubble wrap packaging material (patpat) inside the box indicates that the product has been carefully stored or recently opened.
Physical Deformations and Mechanical Component Analysis
A detailed examination of the device’s casing revealed no scratches, dents, cracks, or breakages. The oxygen outlet nozzle (metal-tipped part) is clean and free of deformation. The device’s air intake/exhaust grilles appear clean, with no dust accumulation observed. The fabric of the carrying bag is robust, with no unravelling or tears. The strap attachments and clips appear intact.
Electronic Components and Screen Status
The device features one LCD screen. As the device is powered off in the visuals, its operational status (presence of pixel errors) cannot be confirmed; however, there are no physical scratches or impact marks on the screen surface. The following buttons are located on the control panel and appear physically intact:
- On/Off button
- (+) and (-) buttons for flow adjustment
- Audible alert (alarm) button
- Volume/Speaker button
Battery and Power Supply
The Inogen Rove 6 model is a portable device powered by a battery. From its form, it is understood that there is a battery structure integrated into the base of the device in the visual; however, as the battery was not externally removed and photographed, precise information regarding its capacity (Standard or Extended) cannot be provided. No swelling or deformation is observed in the battery compartment when viewed from the outside.
Label and Technical Information
The primary legible text on the device consists of brand and model information. Technical details such as serial number (SN), lot number, or REF code are typically found on a label on the bottom or back of the device; however, these sections could not be viewed in the available visuals and therefore could not be reported. No date-containing label, other than the place of manufacture information, was discernible on the box in the visual.
Potential Malfunction Risk and Evaluation
During the visual inspection, no breakages, missing parts, or severe damage that would hinder the device’s operation were detected. The cleanliness of the device and its presentation with its box indicate its excellent condition. No visible malfunction risk is present. However, as it is an electronic medical device, it is always a recommended procedure for battery performance and oxygen purity levels to be checked by an authorized service or technical personnel before commencing active use.