Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 27.09.2025
Respironics BiPAP autoSV Respiratory Device Analysis Report
Product Overview and Contents
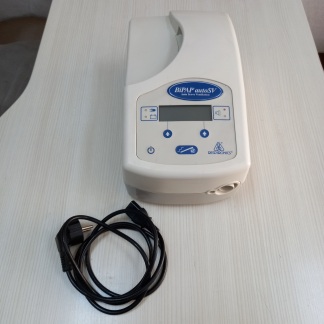
The visuals show a BiPAP (Bilevel Positive Airway Pressure) device and its accessories, understood to be used in the treatment of respiratory disorders such as sleep apnea. The product set consists of a total of 4 parts. These parts are; the main unit, power adapter, power cable, and one smart card.
- Main Device: 1 unit
- Power Adapter: 1 unit
- Power Cable: 1 unit
- Smart Card: 1 unit
Essential components such as the breathing circuit (hose) and mask, which are necessary for the device’s use, are not present in the visuals. This indicates that the device is not ready for standalone use.
Brand, Model, and Originality
The brand of the device shown in the visuals has been clearly identified as Respironics from both the logo on the front face of the device and the text on the smart card. The device’s model is clearly written on its side surface as BiPAP autoSV. Assessing the logo, font characters, and overall craftsmanship quality, it is understood that the product is an original Respironics item.
Intended Use and Functionality
The BiPAP autoSV is an advanced respiratory therapy device used particularly in the treatment of complex and central sleep apnea. The device continuously monitors the patient’s breathing pattern, automatically adjusts pressure support when needed, and thereby helps stabilize respiration. The “Encore Pro” branded smart card in the visual is used for recording patient treatment data and for monitoring by healthcare professionals.
Physical Condition and Cosmetic Evaluation
The general condition of the device is assessed as “used”. Cosmetically, it is in good condition. No deep scratches, cracks, or severe deformations are observed on the casing. There is some dust and minor signs of wear due to use on the surfaces and in recessed areas such as ventilation grilles. The device’s screen is intact and has no cracks. The power adapter and cables included with the device are also in good physical condition.
Technical Details and Accessories
- Main Unit: The device’s front panel features a digital display, control buttons, and an air outlet port. Its rear panel has a power input and two additional sockets, understood to be used for data transfer. The “19.9” figure seen on the screen suggests that the device is operational and likely displays a pressure value.
- Power Adapter: An original Respironics brand power adapter with model number 1015642 is provided with the product. According to its label, the adapter has an output power of 12V and 4.16A.
- Smart Card: The Respironics Encore Pro branded smart card supports the device’s data recording feature.
- Missing Accessories: Essential parts for the device to perform its basic function, such as the breathing tube and patient mask, are not present in the visuals.
Label and Code Information
Readable label information on the device and its accessories is listed below. The label containing the device’s main serial number cannot be clearly discerned in the visuals.
- Label on Device: There is a technical service label on the front face. This label contains the information of the “SESAN” company and the code “Ref: B3/8892/21”.
- Power Adapter Label:
- Brand: RESPIRONICS
- Model No: 1015642
- Output: 12V — 4.16A
Potential Risk Assessment
Based on visual inspections, no obvious malfunction or severe physical damage has been detected on the device. The device’s screen being operational indicates that its basic electronic components are functional. However, as the product is a used medical device, it is recommended that it undergo maintenance and calibration checks by a specialized technical service. The absence of any crushing or wear on the cables suggests that it does not pose an electrical risk in its current condition.